Brief communication |
Peer reviewed |
Evaluating the efficacy of protocols for decontaminating
tail-clipping instruments
Roberta M. Alvarez, DVM; Jason A. Kelly; Sandra F. Amass, DVM, PhD, Diplomate ABVP; Jessica Schneider, BS, RVT; Darryl Ragland, DVM, PhD
Department of Veterinary Clinical Sciences, Purdue University, 1248 Lynn Hall, West Lafayette, IN 47907-1248; E-mail: amasss@purdue.edu.
Alvarez RM, Kelly JA, Amass SF, et al. Evaluating the efficacy of protocols for decontaminating tail-clipping instruments. J Swine Health Prod. 2002;10(5):209-211. Also available as a PDF.
Summary
Research to determine effective protocols for decontaminating pig-processing instruments is lacking. In this study, decontamination of tail-clipping instruments by dipping in a chlorhexidine solution or water was not as effective as wiping the instruments with a clean cloth.
Keywords: swine, disinfection,
decontamination, processing, biosecurity
swine, disinfection,
decontamination, processing, biosecurity
Received: December 17, 2001
Accepted: February 14, 2002
Despite the progress that has been made in many areas of swine pro-duction, no one has published studies addressing neonatal polyarthritis subsequent to pig processing since the mid-1970's, even though it continues to be a problem. In 1975, polyarthritis due to bacterial infections was reported in 3.3% of pigs over 3 days of age.1 Neonatal poly-arthritis is most commonly caused by hemolytic streptococci; however, Staphylococcus spp, Escherichia coli, and many other bacteria may also cause such infections.1,2
Risk factors for neonatal polyarthritis include tail docking
and teeth clipping.1,2 Thus, improper decontamination
of processing instruments may have a significant impact on pig
health. Protocols on many pork production facilities require that
instruments used in processing, such as teeth clippers, tail clippers,
and ear notchers, be decontaminated before each use. However,
strict implementation of instrument sanitation may be difficult.
Often producers neglect such procedures because they are
inconvenient. Moreover, there is no scientific evidence that these
procedures are effective.
We hypothesized that proper decontamination of processing instruments might assist in preventing mechanical transmission of bacteria among pigs during processing. Our premise was that efficacious protocols should eliminate or significantly reduce numbers of bacteria on the processing instrument. For the purposes of this study, disinfection was defined as aerobic culture of less than one viable bacterium per cm2 of instrument cutting surface area.3 The objective of this study was to determine which of the following protocols was most effective at reducing aerobic bacterial contamination of tail-clipping instruments: dipping instruments in a disinfectant solution; dipping instruments in farm tap water; or wiping instruments with a clean cloth.
Materials and methods
Forty 2- to 4-day-old barrows from ten litters were randomly allocated to three treatment groups and one control group of ten pigs each. Each treatment group corresponded to one of three protocols, each performed after clipping the tail of a single pig. Protocol One consisted of dipping tail-clipping instruments for 3 seconds in a freshly prepared solution of 2% chlor-hexidine diacetate (Nolvasan; Fort Dodge Laboratories, Inc, Fort Dodge, Iowa) diluted to 24 mL per liter of sterile water. Protocol Two consisted of dipping tail-clipping instruments for 3 seconds in an unused cup of farm tap water (chlorinated). Protocol Three consisted of wiping tail-clipping instruments with a clean cloth laundered in Surf Ultra laundry detergent (Lever Bros Co, Greenwich, Connecticut). The positive control consisted of sampling untreated tail-clipping instruments that had each been used to clip the tail of a single pig. One individually wrapped, sterile tail-clipping instrument (Supravet; Syrvet, Des Moines, Iowa) per pig was used to cut each pig's tail to a length of approximately 1.9 cm. Ten repetitions were performed for each treatment.
A standardized area (approximately 2 cm2) of each
tail-clipping instrument was sampled using a sterile cotton swab.
The sampled area included the bottom cutting edge and the surface
of the bottom blade that faced the pig's body. The top blade of
the instrument was not sampled. Swab samples were immediately
placed in individual tubes containing 2 mL of chemical broth to
inactivate residual disinfectant
(D/E Neutralizing Broth; Becton-Dickinson, Franklin Lakes, New
Jersey). The swab portion was broken off into the tube of solution
and the contents were mixed by hand agitation. Samples were placed
on cold packs in a cooler on the farm and during transport, then
refrigerated until processing. A 100-L aliquot of the original
sample was plated directly onto 5% blood agar. Additionally, serial
tenfold dilutions of original samples were made using D/E Neutralizing
Broth, and a 100-mL aliquot of each dilution was plated directly
onto 5% blood agar. Samples were incubated for 24 hours at 37°C.
Colonies of aerobic bacteria were counted and total aerobic bacterial
counts per 2 cm2 were calculated.
The resulting bacterial counts did not pass normality tests; therefore, the Kruskal-Wallis test (nonparametric ANOVA) was the statistical test of choice, as it is indicated to compare the medians of three or more groups of non-parametric data sets.4 Dunn's multiple comparisons test was then used to compare the difference in the sum of ranks between two treatments with the expected average difference (based on the number of groups and sample size). For all tests, P<.05 was considered statistically significant. GraphPad InStat version 3.00 for Windows 95 (GraphPad Software, San Diego, California) was used for statistical calculations.
Background contamination control samples were collected. Briefly,
five 3-mL aliquots, respectively, of unused farm tap water and
freshly prepared chlorhexidine diacetate solutions were cultured
as described above, except D/E broth was replaced by sterile water.
Clippers were
exposed to farrowing room air for approx-imately 3 seconds, sampled,
and processed as described above. Additionally, five unused towels
were sampled by exposing the towel to farrowing room air for approx-imately
3 seconds and then placing a 1-cm x 3-cm sample of
towel into sterile water and culturing as described above.
The prevalence of polyarthritis subsequent to treatment was not measured in this study.
Results
Background contamination
The mean aerobic bacterial count (+/- SE) for the five tap water samples was 692 +/- 594 colony forming units (CFU) per mL and ranged from 0 to 3060 CFU per mL. Only one of the five water samples was sterile. All five samples of chlorhexidine diacetate solution were sterile. Four of five towel samples exposed to room air were sterile. The aerobic bacterial count on the fifth towel was 13.33 CFU per 2 cm2. The mean aerobic bacterial count (+/- SE) on unused instruments exposed to room air was 32 +/- 12 CFU per 2 cm2 and ranged from 20 to 80 CFU per 2 cm2.
Bacterial counts on treated instruments
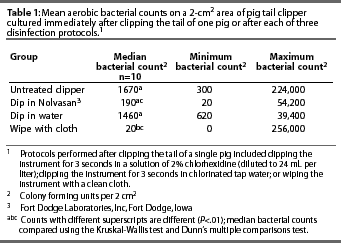
Variation among median aerobic bacterial counts for the treatments was greater than expected by chance (P=.0004). There were no differences between median bacterial counts after dipping the instrument in chlorhexidine diacetate solution for 3 seconds or dipping the instrument in water for 3 seconds, compared to not treating the instrument. Wiping the instrument with a clean cloth reduced the numbers of bacteria cultured (P<.01) compared to not treating the instrument (Table 1).
Discussion
Many farrowing house personnel clean processing instruments after treating each pig to minimize the risk of spreading infections within and among litters. Common procedures include rinsing the instruments with water or dipping them in a solution of disinfectant after processing each pig. All treatments in this study were conducted in the farrowing house of a commercial pork production unit. Consequently, background contamination of tail-clipping instruments and towels was expected and did occur. Additionally, the farm tap water used was contaminated with bacteria and thus may have increased contamination of the tail-clipping instruments in some cases.
The results of this study demonstrated that dipping tail-clipping instruments in water or in a solution of chlorhexidine diacetate, as commonly practiced in many pork production facilities, and as implemented in this study, did not disinfect tail-clipping instruments nor significantly reduce aerobic bacterial counts compared to not treating instruments. Lack of disinfection using chlorhexidine diacetate may have been the result of inadequate contact time. A 10-minute contact time for disinfection of inanimate objects is the label recommendation. A 10-minute contact time was not tested in this study, so the efficaciousness of an extended contact time is unknown and should be tested in future studies. However, producers wishing to test the efficacy of an extended disinfectant contact time for processing instruments can do so by rotating multiple tail-clipping instruments, such that after one instrument is used, it is soaked in disinfect for 10 minutes before that instrument is reused.
These experiments demonstrated that wiping the instruments with a clean cloth between uses was an effective means of reducing aerobic bacterial counts on tail-clipping instruments most of the time. Wiping the tail-clipping instrument with the cloth was the only treatment that achieved disinfection. Disinfection occurred in two of ten replicates using this treatment. However, the highest bacterial count (256,000 CFU per 2 cm2) also resulted from this treatment. In this study, wiping the contaminated blade of the tail-clipping instrument with a clean cloth between uses was the only treatment to significantly reduce aerobic bacterial counts, compared to using untreated instruments. This study was conducted in the farrowing house of a commercial farm, yet four of five towels remained sterile after brief exposure to room air. The towels used in this study were kept wrapped until use to prevent contamination from facility air. Depending on farm sanitation, towels might become increasingly contaminated and negate the beneficial effects of this treatment. Moreover, using the same towel multiple times might prove ineffective. Therefore, the authors recommend that precautions be taken to protect single-use towels or unused portions of multi-use towels on the processing cart by maintaining them in a closed bag or container prior to use.
Implications
- Dipping tail-clipping instruments in chlorhexidine diacetate solution or water after a single use did not significantly reduce the median aerobic bacterial counts on the cutting blade compared to not treating tail clippers.
- Wiping the cutting blade of processing instruments with a clean cloth between uses provided an effective method for reducing the median aerobic bacterial counts on tail-clipping instruments most of the time.
Acknowledgements
This project was funded by the National Pork Board.
References - refereed
1. Nielsen NC, Bille N. Preweaning mortality in pigs. 7. Polyarthritis. Nordisk Veterinaermedicin. 1975;27:529-543.
2. Riising HJ, Nielsen NC. Streptococcal infection in sucking pigs.1. Epidemiological investigations. Nordisk Veterinaermedicin. 1976;28:65-79.
3. Tamasi G. Testing disinfectants for efficacy. Res sci tech Off In Epiz.1995;14(1):75-79.
4. Motulsky H. Comparing three or more means: analysis of variance. Intuitive Biostatistics. New York, New York: Oxford University Press; 1995:255-262.
