Brief communication |
Peer reviewed |
Elimination of porcine reproductive and respiratory syndrome virus from 30 farms by test and removal
Scott A. Dee, DVM, MS, PhD, Diplomate ACVM
Swine Disease Eradication Center, University of Minnesota College of Veterinary Medicine, Room 385 C, 1988 Fitch Avenue, St Paul, MN 55108; Tel: 612-625-4786; Fax: 612-625-1210; E-mail: deexx004@umn.edu.
Cite as: Dee SA. Elimination of porcine reproductive and respiratory syndrome virus from 30 farms by test and removal. J Swine Health Prod. 2004;12(3):129-133..
Also available as a PDF.
Summary
This paper summarizes data from 30 farms that had undergone porcine reproductive and respiratory syndrome virus (PRRSV) elimination using test and removal. Over a subsequent 12-month monitoring period, 28 of 30 farms remained PRRSV-negative. Nucleic acid sequencing of isolates recovered from the re-infected farms identified an unrelated virus.
Received: June
4, 2003
Accepted: October
20, 2003
The process of test and removal (T&R) has been used to eliminate pseudorabies (Aujeszky's disease) virus and Actinobacillus pleuropneumoniae from infected farms.1,2 Earlier field investigations on the use of T&R for the elimination of porcine reproductive and respiratory syndrome virus (PRRSV) have produced promising results; however, only a small number of herds were tested and breeding herd inventories were < 1500 sows.3,4 Elimination of PRRSV by T&R consists of blood testing the entire breeding herd, identifying PRRSV-infected animals using tests both for antibody and virus, and removing positive animals from the farm. The purpose of this paper is to summarize information from 30 herds in which T&R was used to eliminate PRRSV, and to discuss potential modifications of the existing protocol that might enhance efficacy and reduce cost.
Materials and methods
Farm selection
Breeding herds and artificial insemination centers (AICs) positive for PRRSV were eligible for participation in the study if they could fulfill six criteria (Table 1) developed and published in a previous report.3

Test-and-removal protocol
An ELISA serological test was used for detection of PRRSV antibodies (Idexx HerdChek ELISA; Idexx Laboratories, Westbrook, Maine), in combination with a polymerase chain reaction (PCR) serum assay for detection of PRRSV nucleic acid (TaqMan PCR; Perkin-Elmer Applied Biosystems, Foster City, California).3-6 All tests were conducted at the Minnesota Veterinary Diagnostic Laboratory (St Paul, Minnesota). A sample-to-positive (S:P) ratio of >= 0.4 was considered ELISA-positive. The entire breeding herd (sows, boars, and on-site replacement gilts) or the entire boar population in an AIC was blood tested over a 1- to 2-day period. Sera were collected using a different syringe and needle for each animal. Three samples were pooled for PCR testing, and 1-mL aliquots from each animal were stored at -70°C. If a pool tested positive, the individual samples were then tested. Animals that were both ELISA-positive and PCR-positive were considered to be viremic on the day of sampling. Animals that were ELISA-positive and PCR-negative were considered to have been exposed to PRRSV, but not viremic on the day of sampling. Animals that were ELISA-negative and PCR-positive were considered to be acutely infected. Animals with any of these three profiles were removed from the farm within 1 to 2 days upon receipt of the test results and were replaced by PRRSV-naive stock. Finally, animals that were negative on both tests were considered noninfected and remained in the herd.
Following the completion of the T&R protocol, all participating breeding herds and AICs were monitored monthly for 12 months by Idexx HerdChek ELISA, using a sample size capable of detecting at least one positive pig at an estimated prevalence of >= 5% at a 95% confidence limit with an accuracy of +/- 5%. During each monitoring period, a minimum of 50% of the calculated number of sampled animals were randomly selected from naive replacement stock introduced to the herd after initiation of the T&R protocol, while the remaining samples were from randomly selected animals that had originated in the herd. In addition to being tested by ELISA, AIC populations were monitored weekly by PCR testing of pooled semen samples (three samples per pool) on the basis of the 95% confidence-5% accuracy sample size calculation. Breeding herds or AICs were not considered to be PRRSV-negative unless, after the 12-month monitoring protocol was complete, all original PRRSV-positive animals detected in the herd during the T&R process had been removed, and there had been no detection of seroconversion of naive replacement stock.
As positive and negative controls, three PRRSV-positive breeding herds, three PRRSV-positive AICs, three PRRSV-naive breeding herds, and three PRRSV-naive AICs were monitored for a 12-month segment of the project period using the same protocol as for the T&R herds. For inclusion in the study, positive control herds had to fulfill all selection criteria (Table 1), while negative control herds had to fulfill Criteria 3 through 6. The purpose of testing the positive control herds was to demonstrate that spontaneous elimination of PRRSV is a rare event. Intervention strategies for elimination of PRRSV, such as herd closure or other management strategies, were not implemented in positive control herds during the monitoring period.
Results
The project period extended from October 1998 to July 2002. Thirty farms (designated as T&R farms) and six control farms participated. All 36 farms in the study were located in the Midwest United States across nine states. Testing of samples from all 36 farms involved five diagnostic laboratories. Eighteen T&R farms were AICs and 12 were breeding herds, 10 of which were involved in seedstock production. The mean inventory of the 12 T&R breeding herds was approximately 1500 sows (range 300 to 3000). Four (33.3%) had 300 to 1500 sows, seven (58.3%) had 1500 to 2500 sows, and one (8.3%) had 3000 sows. The mean inventory of the 18 T&R AICs was 172 boars (range 75 to 500). Ten AICs (55.5%) had 75 to 100 boars, six (33.3%) had 150 to 200 boars, and two (11.1%) had either 300 or 500 boars. The mean inventory of control breeding herds was approximately 2000 sows (range 1200 to 2400) and the mean inventory of the control AICs was 150 boars (range 50 to 200). None of the 36 farms used PRRSV vaccines prior to or during the study period.
A summary of diagnostic data from each study herd is shown in Tables 2 and 3. On the day of testing, the mean seroprevalence in the 12 T&R breeding herds was 10% (range 5 to 15%) while the mean sero-prevalence in the 18 T&R AICs was 17% (range 10 to 25%). The percentage of animals removed ranged from 2 to 12% in the 12 T&R breeding herds and from 10 to 20% in the 18 T&R AICs. Across all 30 T&R farms, the majority of the removed animals (82 to 95%) were ELISA-positive and PCR-negative. However, animals that were ELISA-positive and PCR-positive (4.4 to 12%) or ELISA-negative and PCR-positive (0 to 5.5%) were detected as well. In herds of 1500 sows or fewer, the average time required to test the herd, receive and interpret the results, and remove the designated animals was 5 working days (range 3 to 7 working days). Herds with 2500 to 3000 sows required 7 to 10 working days due to the large number of animals tested and the volume of samples tested by PCR.
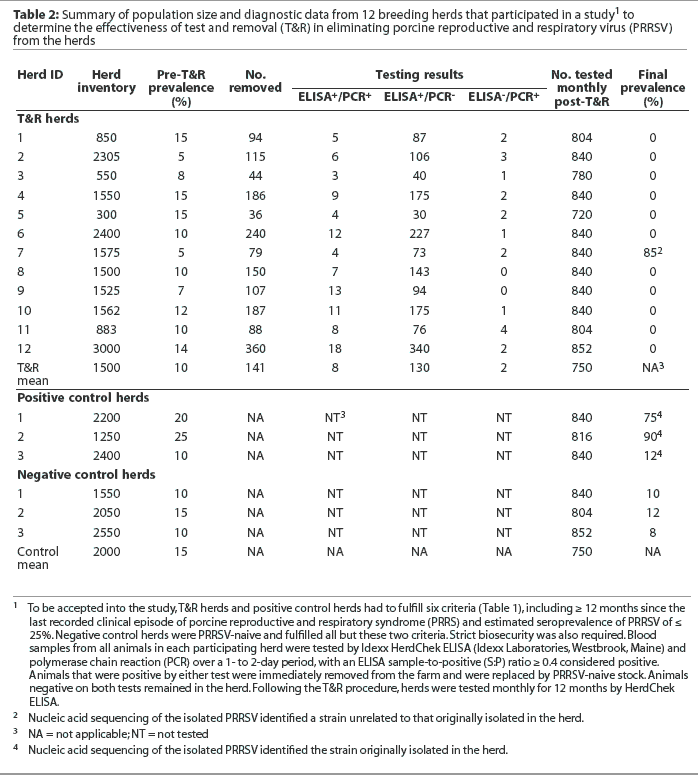
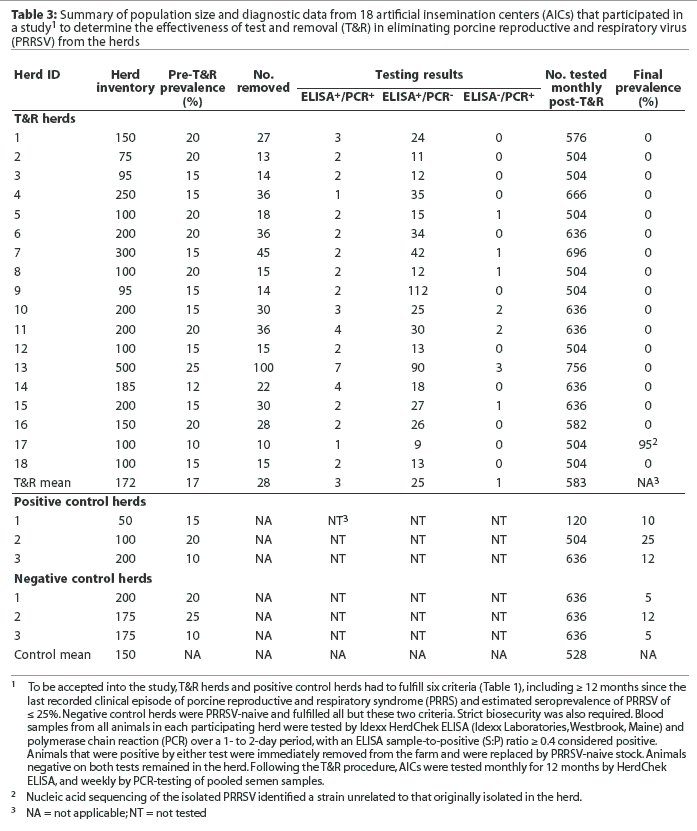
One T&R breeding herd and one AIC became re-infected during their respective monitoring periods; however, in both cases, nucleic acid sequencing of the isolated PRRSV identified a strain unrelated to that originally isolated in the herd (84% and 91% homology, respectively). Throughout the 12-month monitoring period of the control herds, all negative control herds remained negative. All positive control herds remained positive; however, PRRSV seroprevalence increased in two positive control breeding herds, increasing from 20 to 75% in one herd and from 25 to 90% in the other. In each case, PRRSV was isolated, sequenced, and determined to be homologous (> 99%) to the strain that had originally infected the herd.
The diagnostic cost per breeding animal during the whole-herd test was approximately $10 to $12 ($US). The removed nonpregnant sows were slaughtered immediately after results were received; however, whenever possible, pregnant sows were moved to other facilities to gestate and farrow, to minimize production loss.
Discussion
These results indicate that T&R is a highly efficacious, rapid means to eliminate PRRSV from herds that fulfill the selection criteria used in the study. In contrast to elimination of PRRSV by herd closure, which may take 4 to 6 months to complete, the T&R protocol used in this study could be applied immediately upon identification of a qualifying herd. Therefore, T&R required much less time to identify and eliminate at-risk animals in herds that fulfilled the selection criteria. These criteria, developed specifically for well-isolated, endemically infected seedstock herds with excellent biosecurity, required a very low breeding-herd seroprevalence as determined by the Idexx HerdChek ELISA. Therefore, the need to reduce seroprevalence over time, using techniques such as herd closure, was not necessary in any of the study herds.4,7,8 One additional criterion that might have been included was to require a source of PRRSV-naive replacement breeding stock. All herds involved in the study had acquired such a source and had been introducing naive replacement gilts or boars for a 12-month period prior to initiating the T&R protocol.
This study also provided new information on the use of T&R across large numbers of herds monitored over time, efficacy of T&R in large populations, and application of T&R to AICs. The efficacy of T&R was further supported by data from positive control herds. Testing in these herds verified that spontaneous elimination of the virus in large populations is an infrequent event, that PRRSV-seropositive status is likely to be maintained over time, and that recirculation of PRRSV may occur in the absence of intervention strategies to control the disease or eliminate the pathogen.9
Despite these promising results, it may be possible to improve the protocol through the application of new information. A previously recognized limitation of T&R was the inability to distinguish previously exposed animals that have cleared the virus from persistently infected animals. Both groups display a diagnostic profile of ELISA-positive, PCR-negative.3,4 This problem had been further compounded by the questionable validity of using a populations-based diagnostic assay, such as the Idexx HerdChek ELISA, to determine the PRRSV status of individual animals. In this study, testing in 13 of the herds was scheduled to be completed by July 2002, and it was decided to use HerdChek kits to finish the final phase of the monitoring of these herds rather than switch to the new Idexx 2XR ELISA (Idexx Laboratories). Therefore, it is not possible to comment on the role that the Idexx 2XR ELISA might have had on interpretation of data and decisions on whether animals were removed or retained.
It has long been debated whether animals that originate from PRRSV-positive sources but have PRRS ELISA S:P ratios ranging from 0.1 to 0.39 (ie, classified as PRRSV-negative) may be persistently infected. Recent reports from a number of investigators support this hypothesis;10,11 however, one of these studies also demonstrated that sera from such animals are positive by the indirect fluorescent antibody (IFA) test.11 Therefore, it may be feasible to test sera from ELISA-positive, PCR-negative animals by IFA and remove only those that are IFA-positive. While this might potentially reduce the number of removed animals and make T&R a more cost-effective procedure, it must be remembered that the animals in both referenced studies originated from experimentally infected populations, not from field cases. Finally, experienced personnel are required to properly interpret the results of the IFA test, and the test may not detect antibodies to the European strain of PRRSV unless a special assay is performed. However, the relevance of this issue was diminished because selection criterion #3 required molecular sequencing of the open reading frame 5 region of PRRSV isolates recovered from each study herd.
The T&R protocol has other limitations, including the cost of diagnostics, the labor involved in testing large herds, the risk of injury to personnel when testing boars, and the requirement of a population with a low prevalence of seropositive animals. While diagnostic costs could be reduced by eliminating PCR testing of sera, it must be recognized that ELISA-negative, PCR-positive animals were detected in some herds in this study. One option would be to eliminate PCR testing and instead retest the entire population by ELISA 2 to 3 weeks after a herd ELISA test in which seropositive animals are detected. However, the cost and labor involved in another whole-herd test would need to be calculated. In the case of farms in which breeding-herd seroprevalence is > 25%, herd closure might be employed, a protocol that has been well documented.4,7,8 However, it must be remembered that the characteristics of the herds selected for this study included very low breeding herd seroprevalence, isolation from other swine herds, a source of PRRSV-naive seedstock, and no clinical signs of PRRS for a 12-month period. These characteristics most likely played a major role in the success of the T&R procedure, and should be deemed essential when herds are evaluated for potential use of T&R as a PRRSV-elimination strategy.
As of this writing, this is the largest study in the literature summarizing results of a standardized protocol of PRRSV elimination in the field over an extended period of time. However, as for other methods, it appears that the challenge is not limited to eliminating the virus from the population, but rather keeping the herd negative over time. Therefore, swine practitioners must continue to emphasize maintenance of strict herd biosecurity protocols to reduce this ever-present risk.
Implications
- Test and removal is a highly efficacious, rapid means to eliminate PRRSV in selected breeding herds or artificial insemination centers.
- Criteria for selection of herds for test and removal should include low seroprevalence, isolation from other swine herds, a source of PRRSV-negative replacement animals, and no clinical signs of PRRS for a 12-month period.
- Other large-scale efforts utilizing other protocols for elimination of PRRSV should be attempted for comparison with results of the standardized protocol reported here.
References
1. Thawley DG, Gustafson DP, Beran GW. Swine Pseudorabies Eradication Guidelines. 1st ed. Madison, Wisconsin: Livestock Conservation Institute; 1982;1-11.
2. Nielsen R, Thomsen AD, Vesterlund SD. Pleuropneumonia caused by Haemophilus parahaemolyticus: An attempt to control the disease at two progeny testing stations by serological blood testing followed by removal of the seropositive animals and their litter mates. Nord Vet Med. 1976;28:349-352.
3. Dee SA, Bierk MD, Molitor TW. An evaluation of Test and Removal for the elimination of PRRS virus from 5 swine farms. Can J Vet Res. 2001;65: 22-27.
4. Dee SA, Molitor TW, Rossow KD. Epidemiological and diagnostic observations following elimination of PRRS virus using Test and Removal. Vet Rec. 2000;146:211-213.
*5. Snyder ML, Mermer B, Anderson PR, Wensvoort G, Hill HT. Evaluation data for an immunological ELISA for PRRS. Proc 2nd Int Symp PRRSV. Copenhagen, Denmark. 1995:15.
*6. Molitor TW, Tune KA, Shin J, Collins JE, Kapur V. Application of TaqMan PCR in the detection of porcine reproductive and respiratory syndrome virus. Proc Allen D. Leman Swine Conf. St Paul, Minnesota. 1997;173-175.
7. Dee SA, Joo HS, Pijoan C. Controlling the spread of PRRS virus in the breeding herd through management of the gilt pool. Swine Health Prod. 1994;3:64-69.
*8. Torremorell M, Christianson WT. PRRS Eradication. Proc 1st Int Symp Swine Dis Erad. Minneapolis, Minnesota. 2001:7-10.
9. Freese WR, Joo HS. Cessation of PRRS virus spread in a commercial swineherd. Swine Health Prod. 1993; 2:13-15.
10. Horter DC, Pogranichniy RM, Chang CC, Evans RB, Yoon KJ, Zimmerman JJ. Characterization of the carrier state of porcine reproductive and respiratory syndrome virus infection. Vet Microbiol. 2002;86:213-228.
11. Batista L, Dee SA, Rossow KD, Olin M, Molitor TW, Xiao Z, Murtaugh MP, Pijoan C. Detection of porcine reproductive and respiratory syndrome virus in animals with low positive or negative ELISA sample-to-positive ratios. Vet Rec. 2004;154: 25-26.
*Non-refereed references.
