Original research |
Peer reviewed |
Effects of adjuvants on porcine circovirus type 2-associated lesions
Efectos de los adyuvantes en las lesiones asociadas con el circovirus porcino tipo 2
Effets des adjuvants sur les lésions associées au circovirus porcin de type 2
Marlin J. Hoogland, DVM, MS; Tanja Opriessnig, Dr med vet; Patrick G. Halbur, DVM, PhD
Department of Veterinary Diagnostic and Production Animal Medicine, College of Veterinary Medicine, Iowa State University, Ames, Iowa. Corresponding author: Dr P. G. Halbur, Department of Veterinary Diagnostic and Production Animal Medicine, College of Veterinary Medicine, Iowa State University, Ames, IA 50011; Tel: 515-294-1137; Fax: 515-294-3564; E-mail: pghalbur@iastate.edudeexx004@umn.edu.
Cite as: Hoogland MJ, Opriessnig T, Halbur PG. Effects of adjuvants on porcine circovirus type 2-associated lesions. J Swine Health Prod. 2006;14(3):133-139.
Also available as a PDF.
SummaryObjective: To determine if adjuvants differ in their ability to trigger lesions associated with porcine circovirus type 2 (PCV2). Methods: Ninety pigs randomly assigned to five groups were vaccinated intramuscularly at 4 and 6 weeks of age with 2 mL of a commercial Mycoplasma hyopneumoniae (M hyo) vaccine with oil-in-water adjuvant (Group 1), a commercial M hyo vaccine with an aqueous-carbopol adjuvant (Group 2), an experimentally produced M hyo vaccine with an oil-in-water adjuvant (Group 3), or an experimentally produced M hyo vaccine with an aluminum hydroxide adjuvant (Group 4), or were sham-vaccinated with saline (Group 5). All pigs were inoculated intranasally at 6 weeks of age with PCV2 (Day 0). Half of the pigs were necropsied at Day 21 and the remaining pigs at Day 35. Results: No clinical disease was observed in any pigs during this study. At Day 21, lymphoid depletion was more severe in all M hyo-vaccinated pigs than in the saline-treated pigs (P < .05). At Day 35, greater amounts of PCV2 DNA were found in serum, more severe lymphoid lesions were observed, and more PCV2 antigen was detected in lymphoid tissues in Groups 1 and 3 (oil-in-water adjuvant) compared to Groups 2, 4, and 5 (P < .05). Implications: Under the conditions of this study, oil-in-water adjuvanted vaccines are more likely to enhance PCV2-associated lesions than aqueous-carbopol or aluminium hydroxide adjuvanted vaccines. Practitioners must weigh benefits and efficacy of vaccines intended to control coinfections against the potential negative effect certain vaccines may have on PCV2 replication. | ResumenObjetivo: Determinar si los adyuvantes difieren en su habilidad para desencadenar las lesiones asociadas con el circovirus porcino tipo 2 (PCV2 por sus siglas en inglés). Métodos: Noventa cerdos asignados al azar a cinco grupos diferentes fueron vacunados intramuscularmente a las 4 y 6 semanas de edad con 2 mL de una vacuna comercial de Mycoplasma hyopneumoniae (M hyo por sus siglas en inglés) con un adyuvante de aceite en agua (Grupo 1), una vacuna comercial de M hyo con un adyuvante de carbopol acuoso (Grupo 2), una vacuna de M hyo producida experimentalmente con un adyuvante de aceite en agua (Grupo 3), con una vacuna de M hyo producida experimentalmente con un adyuvante de hidróxido de aluminio (Grupo 4), o fueron inoculados con solución salina (Grupo 5). Todos los cerdos fueron inoculados intranasalmente a las 6 semanas de edad con PCV2 (Día 0). El Día 21, la mitad de los cerdos fueron sacrificados y se les realizó una necropsia; el resto de los cerdos fueron sacrificados el Día 35. Resultados: No se observó enfermedad clínica en ninguno de los cerdos durante este estudio. En el Día 21, la depleción linfoide fue más severa en todos los cerdos vacunados con M hyo comparada con los cerdos inoculados con el solución salina (P < .05). En el Día 35, se encontró una mayor cantidad de DNA de PCV2 en suero, se observaron lesiones linfoides más severas y se detectó más antígeno de PCV2 en los tejidos linfoides en los Grupos 1 y 3 (adyuvante de aceite en agua) en comparación con los Grupos 2, 4, y 5 (P < .05). Implicaciones: Es más probable que las vacunas que contienen un adyuvante de aceite en agua aumenten las lesiones asociadas con PCV2 que las vacunas con carbopol acuoso o hidróxido de aluminio. Los veterinarios deben sopesar los beneficios y la eficacia de las vacunas utilizadas para controlar los efectos de las coinfecciones contra el potencial efecto negativo que ciertas vacunas pueden producir con la replicación de PCV2. | ResuméObjectif: Déterminer si des adjuvants diffèrent dans leur capacité à initier des lésions associées avec le circovirus porcin de type 2 (PCV2). Méthodologie: Quatre-vingt-dix porcs répartis de manière aléatoire en cinq groupes ont été vaccinés par voie intramusculaire à 4 et 6 semaines d'âge avec 2 mL d'une des préparations suivantes: un vaccin commercial contre Mycoplasma hyopneumoniae (M hyo) avec un adjuvant de type huile dans l'eau (Groupe 1), un vaccin commercial contre M hyo avec un adjuvant de type carbopol aqueux (Groupe 2), un vaccin anti-M hyo expérimental avec un adjuvant huile dans l'eau (Groupe 3), un vaccin anti-M hyo expérimental avec un adjuvant à base d'hydroxyde d'aluminium (Groupe 4), ou de la saline (Groupe 5). Tous les porcs ont été inoculés par voie intranasale à 6 semaines d'âge avec du PCV2 (Jour 0). La moitié des porcs ont été soumis à une nécropsie au Jour 21 et les porcs restants au Jour 35. Résultats: Aucun signe clinique n'a été noté chez les porcs au cours de cette étude. Au Jour 21, la déplétion des tissus lymphoïdes était plus sévère chez tous les porcs vaccinés contre M hyo que chez les animaux vaccinés avec de la saline (P < .05). Au Jour 35, de plus grandes quantités d'ADN de PCV2 ont été retrouvées dans le sérum, des lésions aux tissus lymphoïdes plus sévères ont été observées, et plus d'antigène du PCV2 ont été détectés dans les tissus lymphoïdes des Groupes 1 et 3 (adjuvant huile dans l'eau) comparativement aux groupe 2, 4, et 5 (P < .05). Implications: Les vaccins avec des adjuvants de type huile dans l'eau sont plus susceptibles d'accentuer les lésions associées au PCV2 que les adjuvants à base de carbopol aqueux ou d'hydroxyde d'aluminium. Les praticiens doivent donc prendre en considération les bénéfices et l'efficacité des vaccins pour contrôler les co-infections versus les effets négatifs potentiels que certains vaccins peuvent avoir sur la réplication de PCV2. |
Keywords: swine, immunostimulation,
porcine circovirus type 2, adjuvants, postweaning multisystemic wasting syndrome
Search the AASV web site
for pages with similar keywords.
Received: November
15, 2004
Accepted: May
23, 2005
Diseases associated with porcine circovirus type 2 (PCV2) have recently become a major global problem. This virus is thought to be the causative agent of postweaning multisystemic wasting syndrome (PMWS), plays a role in the porcine respiratory disease complex,1 and is associated with sporadic abortions.2 Postweaning multisystemic wasting syndrome is characterized clinically by signs of wasting and pneumonia. Microscopically, PMWS is characterized by lymphoid depletion of follicles in lymphoid tissue, with variable degrees of granulomatous inflammation in a variety of organs and demonstration of PCV2 antigen or nucleic acids associated with these lesions.3 Numerous investigators have demonstrated that PMWS is triggered in a high percentage of pigs when PCV2-infected pigs are co-infected with other pathogens such as porcine parvovirus,4-7 porcine reproductive and respiratory syndrome virus (PRRSV),8-10 and Mycoplasma hyopneumoniae (M hyo).11 Several groups have experimentally reproduced the hallmark lesions of lymphoid depletion after singular PCV2 infection, but were either unable to reproduce PMWS4,10,12 or induced a lower incidence of PMWS compared to that observed in the co-infection models.5,6,13,14 Harms et al (2001)8 and Bolin et al (2001),15 however, were able to induce clinical disease consistent with PMWS in a high percentage of cesarean-derived, colostrum-deprived piglets infected with PCV2 alone.
Krakowka et al (2000)4 were the first to demonstrate induction of PMWS in gnotobiotic pigs that had been singularly infected with PCV2 after administration of keyhole limpet hemocyanin in Freund's incomplete adjuvant (KLH-IFA), thus hypothesizing the need for immune stimulation in the expression of PMWS. However, recent work with KLH-IFA by Ladekjær-Mikkelsen et al (2002),16 using specific-pathogen-free piglets inoculated intranasally with PCV2, demonstrated that both immunostimulated and nonimmuno-stimulated piglets infected with PCV2 developed PMWS. Furthermore, Resendes et al (2004)17 compared conventionally raised, PCV2-infected groups of pigs that did or did not receive vaccine adjuvant and concluded that the principle of immuno-stimulation as a cause of enhanced lesions or diseases associated with PCV2 may not be applicable in all cases, and that not all adjuvants used in commercial vaccines are capable of triggering mechanisms for PMWS development.
On the contrary, field trials18-20 and experimental studies21,22 suggest that use of common adjuvanted commercial vaccines may enhance the severity of PCV2-associated disease. On the basis of this information, some veterinarians may advise their clients to discontinue use of commercial vaccines in herds with recurrent PMWS and respiratory disease associated with PCV2. This may minimize the risk of triggering progression of PCV2 infection to PCV2-associated disease, but it may also allow for recrudescence of other diseases (eg, respiratory disease caused by M hyo) that are generally effectively controlled by vaccination. More information is needed to guide veterinarians and producers in selecting vaccines that could be used or should be avoided in herds with PCV2-associated diseases.
We hypothesized that one or more of the commercial adjuvants enhances PCV2 replication and by doing so, triggers progression of PCV2 infection to induce lymphoid depletion and PMWS. We hypothesized that commercial adjuvants are sufficiently distinct chemically that variability in the extent of their effect on PCV2 replication and PCV2-associated lesions should be measurable in vivo. Our objective was to compare the degree of enhancement of PCV2-associated lesions induced by different types of adjuvants commonly used in commercially available vaccines.
Materials and Methods
Animal source
Ninety segregated-early-weaned pigs from a high health-status herd, shown to be PRRSV and M hyo-free on the basis of monthly serological monitoring, were weaned at 12 to 14 days of age and brought to the Livestock Infectious Disease Isolation Facility at Iowa State University, Ames, Iowa. Piglets chosen for the experiment were selected from sows with undetectable or low PCV2 antibody levels 2 weeks prior to farrowing, as measured by an enzyme-linked immuno-sorbent assay (ELISA)23 based upon PCV2 open-reading frame 2 (ORF2) (ie, pigs with sample-to-positive [S:P] ratios < 0.5). All protocols for this experiment were approved by the Iowa State University Animal Care Committee.
Experimental design
The experimental design is summarized in Table 1. Blood samples were collected from the piglets at 2 weeks of age upon arrival at the research facility, and serum was tested by the same PCV2 ELISA as was used for the sows. The piglets were blocked by ELISA S:P ratio and randomly assigned to five treatment groups of 17 to 19 animals per group. Pigs were randomly assigned to housing in two rooms, with eight pens per room and six pigs per pen. All pigs were either vaccinated or sham-vaccinated at 4 and 6 weeks of age. Pigs in Groups 1 and 2 were vaccinated with one of two commercially available vaccines following the manufacture's recommendation, pigs in Groups 3 and 4 were vaccinated with one of two experimentally produced vaccines, and pigs in Group 5 were sham-vaccinated with saline. Pigs in all groups received 2 mL of a designated vaccine or saline intramuscularly behind the ear in the neck region at 4 weeks (right neck) and 6 weeks of age (left neck). Two weeks after the first injection, on the day they received the second injection, all pigs were inoculated with PCV2 (Day 0). After PCV2 inoculation, pigs were weighed weekly, clinical observations were recorded daily, and rectal temperatures were recorded on alternate days. Blood samples were collected before vaccination, before inoculation, and at Days 7, 14, 21, 28, and 35. Half of the pigs were necropsied at Day 21 and the remaining pigs at Day 35, and gross and microscopic lesions were assessed.
Table 1: Adjuvant types and vaccines administered to pigs experimentally infected with porcine circovirus type 2 (PCV2)*
* 2 mL of Mycoplasma hyopneumoniae vaccine or saline was administered intramuscularly behind the ear at 4 weeks (right neck; Day -14) and 6 weeks (left neck; Day 0). Pigs were inoculated intranasally with PCV2 on Day 0 and necropsied on Day 21 (9 weeks of age) or Day 35 (11 weeks of age). † Pfizer Animal Health Inc, New York, New York. ‡ Fort Dodge Animal Health Inc, Fort Dodge, Iowa. § Experimental vaccines formulated for Groups 3 and 4 contained both the same volume and type of M hyopneumoniae antigen and the same amount of adjuvant as Suvaxyn RespiFend MH. |
Vaccines and adjuvants
Four M hyo vaccines were used in this experiment (Table 1). The vaccine used in Group 1, RespiSure (Pfizer Animal Health Inc, New York, New York), contains the proprietary oil-in-water adjuvant Amphigen. The vaccine used in Group 2, Suvaxyn RespiFend MH (Fort Dodge Animal Health Inc, Fort Dodge, Iowa), contains a proprietary carbopol-based aqueous adjuvant. Experimental vaccines were formulated for Groups 3 and 4 using the same volume and type of M hyo antigen contained in Suvaxyn RespiFend MH, and amounts of adjuvant equivalent to that contained in Suvaxyn RespiFend MH. Thus, the vaccines used in Groups 2, 3, and 4 differed only in the type of adjuvant used.
Virus inoculum
The PCV2 isolate used in the study was isolate ISU-40895, which was originally obtained from a pig with PMWS in a western Iowa herd experiencing moderate losses due to PMWS.24 Virus was propagated in PK-15 cells free of PCV1 and PCV2, as previously described.25 Each pig received 5 mL of virus suspension intranasally, containing the eighth passage of 104.5 median tissue culture infectious doses (TCID50).
Clinical scoring
Severity of respiratory disease was estimated and recorded using a scoring system that ranged from 0 to 6 (0 = normal; 6 = severe dyspnea and abdominal breathing) as previously described.23 Clinical signs of diarrhea, icterus, and pallor were estimated (0 = normal; 1 = mild; 2 = moderate; 3 = severe) and recorded.
Serological testing
A PCV2 ELISA, based on recombinant ORF2 caspid protein of PCV2, was performed on serum samples as previously described.23 Samples with S:P ratio >= 0.2 were considered positive for PCV2-specific antibodies.
Evaluation of gross and microscopic lesions
At necropsy, macroscopic lung lesions (percent of lung involvement)26 and size of lymph nodes, scored from 0 (normal) to 3 (three times normal size), were estimated by a veterinary pathologist (PGH). Tissue samples were collected from lymph nodes (superficial inguinal, external iliac, mediastinal, tracheobronchial, and mesenteric), lung, liver, spleen, tonsil, kidney, ileum, and colon, placed in 10% neutral buffered formalin, and routinely processed for microscopic examination. Lungs were insufflated with formalin at the time of necropsy. Microscopic lesions were evaluated in a blinded fashion. Lymphoid depletion of follicles in lymph nodes, tonsil, and spleen were scored from 0 (none) to 3 (severe). Histiocytic replacement of follicles was also scored from 0 (none) to 3 (severe). Lesions in the liver, kidney, and heart were also scored from 0 (normal) to 3 (severe lympho-histiocytic inflammation).21
Immunohistochemistry
Sections of lymph nodes (superficial inguinal, external iliac, mediastinal, tracheobronchial, and mesenteric), tonsil, and spleen were examined by immunohistochemistry (IHC), as described previously,27 to detect and quantify PCV2 antigen. The amount of PCV2 antigen was scored in a blinded fashion from 0 (none) to 3 (strongly positive).
DNA extraction and quantitative PCR
DNA was extracted weekly from serum from all pigs using a commercial DNA isolation kit (QIAamp DNA Blood Mini Kit; Qiagen, Valencia, California). DNA extracts were then used to quantify the amount of PCV2 DNA by real-time PCR as previously described.21
Statistical analysis
For parametric data (including serological results, PCR, animal temperature, and animal weights), analysis of variance (ANOVA) was used to determine differences among groups. If significant differences were identified (P < .05), pairwise testing using Tukey-Kramer adjustment was then per formed. For nonparametric data (including respiratory scores and microscopic and immunohistochemistry scores), a Kruskal-Wallis ANOVA test was performed to determine differences among groups. If significant differences were identified (P < .05), pairwise Wilcoxon tests were performed. A 95% confidence interval was used to reject the null hypothesis.
Results
Clinical disease and gross lesions
No clinical signs of disease were observed in any groups for the duration of this study. Therefore, clinical scores of respiratory disease, diarrhea, icterus, and pallor did not differ among treatment groups (P > .05). In addition, weight gain, daily rectal temperature, and severity of gross lesions did not differ among groups (P > .05).
ELISA results
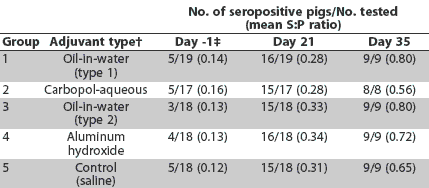
ELISA results are summarized in Table 2. Maternal antibodies had declined (S:P < 0.2) in most pigs by Day 0. Three to five pigs in each group had low levels of maternal antibody (S:P <= 0.3) at the time of inoculation. By Day 21, most pigs were seropositive for PCV2. By Day 35, all pigs remaining in the experiment were seropositive. Incidence of seropositive pigs or the group mean PCV2 S:P ratio did not differ among groups (P >.05).
Table 2: Anti-porcine circovirus type 2 (PCV2) antibodies* in pigs inoculated with PCV2 at 6 weeks of age (Day 0)
* Antibodies detected by ELISA using as antigen open reading frame 2 (ORF2) of PCV2 (Nawagitgul et al).23 A sample-to-positive (S:P) ratio >= 0.2 was considered positive. Blood samples were collected before inoculation and on Days 21 (9 weeks of age) and 35 (11 weeks of age). † Vaccines and adjuvants are described in Table 1. ‡ Positive S:P ratios <= 0.3 due to maternal antibodies. |
Microscopic lesions and immuno-histochemistry
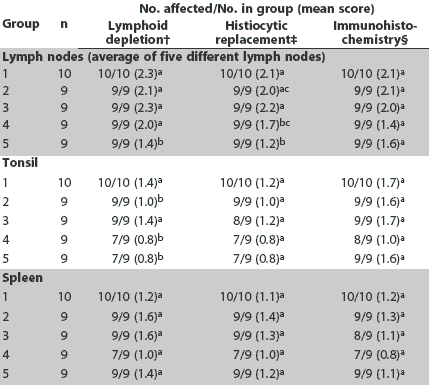
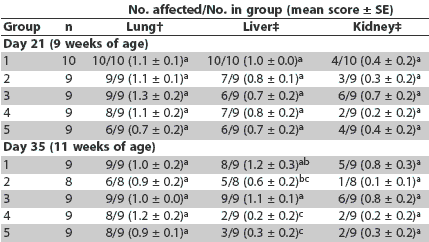
Day 21. More severe lymphoid depletion of lymph nodes was observed in all vaccinated groups than in the saline group; however, vaccinated groups did not differ in this respect (Table 3). More severe histiocytic replacement of follicles was observed in the lymph nodes in Groups 1 and 3 compared to Groups 4 and 5 (P < .05), and more severe lymphoid depletion of the tonsil was observed in Groups 1 and 3 compared to Groups 2, 4, and 5 (Table 3). There were no differences among groups in lung, liver, and kidney lesions (Table 4).
Table 3: Incidence of lesions and mean scores for microscopic lesions and immunohistochemistry in lymph nodes, tonsil, and spleen at Day 21 in pigs vaccinated at 4 and 6 weeks of age with one of four different Mycoplasma hyopneumoniae vaccines* and inoculated with PCV2 at the time of the second injection (Day 0)
* Vaccines and adjuvants described in Table 1. † Lymphoid depletion: score 0 = none; 3 = severe. ‡ Histiocytic replacement of follicles: score 0 = none; 3 = severe. § PCV2 antigen detected by immunohistochemistry: score 0 = none; 3 = severe. abc Within a column, mean group scores with no common superscript are significantly different (Wilcoxon test; P < .05). |
Table 4: Incidence of lesions and mean scores for microscopic evaluation of lung, liver, and kidney at Days 21 and 35 in pigs vaccinated at 4 and 6 weeks of age with one of four different Mycoplasma hyopneumoniae vaccines* and inoculated with PCV2 at the time of the second injection (Day 0)
* Vaccines and adjuvants described in Table 1. † Interstitial pneumonia: score 0 = none; 6 = severe. ‡ Lymphohistiocytic inflammation: score 0 = none; 3 = severe. abc Within a column, mean group scores with no common superscript are significantly different (Wilcoxon test; P < .05). |
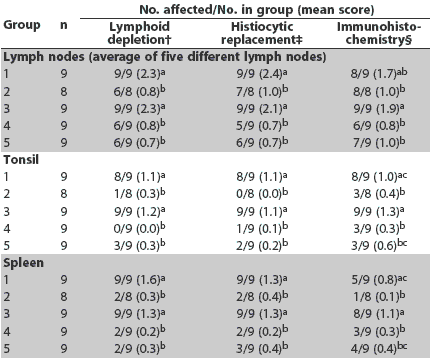
Day 35. Severity of lymphoid depletion in the lymph nodes, tonsil, and spleen was greater in Groups 1 and 3 compared to Groups 2, 4, and 5 (Table 5). Greater amounts of PCV2 antigen were identified in lymph node, tonsil, and spleen in Group 3 compared to Groups 2, 4, and 5 (Table 5). Greater amounts of PCV2 antigen were identified in tonsil and spleen in Group 1 compared to Groups 2 and 4 (Table 5). Severity of histiocytic replacement of follicles in tonsil, spleen, and lymph nodes was greater in Groups 1 and 3 compared to Groups 2, 4, and 5 (Table 5). Severity of lymphohistiocytic hepatitis was greater in Group 1 compared to Groups 4 and 5, and in Group 3 compared to Groups 2, 4, and 5 (Table 4). Overall, severity of lymphoid lesions declined over time, but their duration was prolonged in Groups 1 and 3 compared to Groups 2, 4, and 5.
Table 5: Incidence of lesions and mean scores for microscopic lesions and immunohistochemistry in lymph nodes, tonsil, and spleen at Day 35 in pigs vaccinated at 4 and 6 weeks of age with one of four different Mycoplasma hyopneumoniae vaccines* and inoculated with PCV2 at the time of the second injection (Day 0)
* Vaccines and adjuvants described in Table 1. † Lymphoid depletion: score 0 = none; 3 = severe. ‡ Histiocytic replacement of follicles: score 0 = none; 3 = severe. § PCV2 antigen detected by immunohistochemistry: score 0 = none; 3 = severe. abc Within a column, mean group scores with no common superscript are significantly different (Wilcoxon test; P < .05). |
Detection of PCV2 nucleic acids by PCR
Nucleic acids of PCV2 were detected in all groups by quantitative real-time PCR as early as Day 7. Initial residual analysis of the raw data indicated heterogeneity of variances. This was corrected by log-transforming the raw data. Group differences were not observed until Day 35. At Day 35, more copy numbers of PCV2 genomic DNA were detected in the serum of Group 1 (log mean 5.35 +/- 0.29) and Group 3 (log mean 4.81 +/- 0.43) compared to Group 2 (log mean 4.05 +/- 0.27), Group 4 (log mean 4.26 +/- 0.15), and Group 5 (log mean 4.08 +/- 0.30) (P < .05).
Discussion
The objective of this study was to determine whether adjuvants (as opposed to antigens) in commercial swine vaccines enhance replication of PCV2 and induce a higher incidence of PCV2-associated lesions. Allan et al (2000)22 demonstrated that PMWS occurred in 21% of a group of colostrum-fed, PCV2-infected pigs vaccinated with commercial M hyo and Actinobacillus pleuro-pneumoniae vaccines, compared to 0% in an unvaccinated group. Similarly, field studies conducted by Allan et al (2001)18 revealed that losses attributed to PMWS were higher in vaccinated pigs in two of five groups. In a field trial, Kyriakis et al (2002)19 found that 43% of pigs vaccinated with M hyo (RespiSure) developed PMWS, compared to 11% of those sham-vaccinated with saline. These studies provide evidence not only that PCV2 may require a triggering mechanism, but also that commercial vaccines are capable of triggering PCV2-associated diseases via immune stimulation, which enhances PCV2 replication and increases the incidence of clinical PMWS. Although the results of many studies18,19,21,22 support the association of adjuvanted vaccines with increased incidence and severity of PCV2-associated diseases and lesions, very little work has been done to fully understand what components of any vaccine, and particularly M hyo bacterins, contribute to enhanced PCV2 replication and increased severity of PCV2-associated lesions.
Recently, studies have suggested that not all adjuvanted vaccines play a role in PCV2-associated lesions. Resendes et al (2004),17 using four groups of pigs (a control group, a vaccine-adjuvant group, a PCV2-infected group, and a group receiving both the vaccine adjuvant and PCV2 infection), found that no differences among groups were associated with use of a vaccine adjuvant. However, only one commercial vaccine adjuvant was used in that study.17 On the contrary, Vanderstichel and Hurnik (2003)20 conducted a field trial in herds previously suffering from PMWS. Using different adjuvanted M hyo products (two with oil-in-water adjuvants and one with an aluminum hydroxide adjuvant), they found a higher proportion of pigs treated with the aluminum hydroxide adjuvant developed PMWS compared to pigs treated either with saline or the oil-in-water adjuvant. They concluded that the immunostimulation induced by the aluminum hydroxide adjuvant may have facilitated development of clinical disease. However, neither length of viremia, severity of lesions, nor amount of antigen in lymphoid tissue were established in that study.20 Furthermore, in field studies, it is often difficult to control and account for confounding factors such as concurrent disease.
With the great diversity and chemical variation existing among commercial adjuvants, it is not unexpected that M hyo vaccines vary in effect on PCV2-associated lesions. However, most experimental work to date has focused on use of a single adjuvanted vaccine either triggering or not triggering PCV2-associated lesions and disease. To our knowledge, ours is the first controlled study to compare multiple adjuvanted vaccines. We tested multiple adjuvanted M hyo bacterins, both commercially available and experimentally formulated, administered on a schedule consistent with common practices in the US swine industry (4 and 6 weeks of age) and in a controlled environment. To ensure that differences among groups were not due to administration of different volumes of adjuvant, equal amounts of adjuvant were included in vaccines for Groups 2, 3, and 4. Likewise, to control for a potential M hyo-antigen effect and ensure that differences among groups were due to the type of adjuvant, both of the experimental vaccines (Groups 3 and 4) were identical to the commercial vaccine (Group 2) in both volume and type of M hyo antigen. Thus, only the type of adjuvant differed in the vaccines for Groups 2, 3, and 4. Vicca et al (2003)28 found that variation in virulence exists among M hyo strains. In order to address the role that M hyo antigen may play in PCV2 replication, it was necessary to control the M hyo antigen among Groups 2, 3, and 4 and to compare the response among these groups with that of Group 1, so that differences among these groups could be attributed to the type of adjuvant. Group 1 was included in the study for comparison of two commercial M hyo vaccines and two oil-in-water adjuvanted vaccines, and the sham-vaccinated pigs (Group 5) were included to control for stress associated with the vaccination procedure.
In the early stages of infection (Day 21), the severity of lymphoid depletion associated with PCV2 was greater in vaccinated pigs than controls for all adjuvants tested. In the later stages of infection (Day 35), severity of lymphoid depletion was greater in groups treated with the oil-in-water adjuvants (Groups 1 and 3) compared to the groups treated with aqueous-carbopol and aluminum hydroxide adjuvants.
The RespiSure product used in this study contains a proprietary oil-in-water adjuvant (Amphigen), which no doubt is somewhat different from the oil-in-water adjuvant in the experimental vaccine. However, the experimental oil-in-water adjuvanted vaccine was intentionally produced to mimic the RespiSure vaccine used in Group 1. The results obtained from both oil-in-water products were very similar. This suggests that different types of adjuvants differ in their mechanisms of action, their effects on the immune system, and the body's response to their presence in vaccines. Therefore, it should be expected that different adjuvanted M hyo vaccines contribute to different degrees of severity of PCV2-associated lesions.
The mechanism of the complex interaction of PCV2 and oil-in-water adjuvants is unknown. Because both oil-in-water adjuvanted bacterins produced very similar results despite their different origins and potentially different molecular makeup, veterinarians should explore alternatives to oil-in-water adjuvanted M hyo bacterins in herds suffering from recurrent PCV2-associated diseases. Since PCV2 is a ubiquitous pathogen, biological companies and regulatory agencies should also consider the effects of adjuvants on PCV2 replication when formulating and approving new products.
This study provides important information concerning use of M hyo bacterins in herds suffering from PCV2-associated diseases. Veterinarians must weigh the risks of not using M hyo bacterins, thereby potentially allowing co-infections (ie, with M hyo) to enhance PCV2-associated diseases, against the risk of using these bacterins on PCV2-infected pigs, thereby potentially increasing the severity of PCV2-associated diseases. Different variables must be considered in each herd when vaccine programs are implemented. The information obtained in this study will be immediately useful to veterinarians and practitioners struggling to control PCV2-associated diseases.
Implications
- Under the conditions of this study, oil-in-water adjuvanted vaccines are more likely to enhance PCV2-associated diseases than aqueous-carbopol or aluminium hydroxide adjuvanted vaccines.
- In herds where disease associated with PCV2 has occurred, practitioners must weigh the benefits and efficacies of different vaccines to control co-infections against the potential negative effect certain vaccines may have on PCV2 replication.
- Biological companies and regulatory agencies should consider the effects of adjuvants on PCV2 replication when formulating and approving new products.
Acknowledgements
A special thanks to the American Association of Swine Veterinarians for providing financial support for this project in the form of a summer student research scholarship. The animal and laboratory work was funded in part by the Iowa Livestock Health Advisory Council and in part by Fort Dodge Animal Health, Inc. The authors thank Drs S. Yu, E. L. Thacker, M. Fenaux, and X. J. Meng for providing the PCV2 inoculum for the experiment and Pete Thomas for assistance with the animal work.
References
1. Harms PA, Halbur PG, Sorden SD.Three cases of porcine respiratory disease complex associated with porcine circovirus type 2 infection. Swine Health Prod. 2002;10:27-30.
2. West KH, Bystrom JM, Wojnarowicz C, Shantz N, Jacobson M, Allan GM, Haines DM, Clark EG, Krakowka S, McNeilly F, Konoby C, Martin K, Ellis JA. Myocarditis and abortion associated with intrauterine infection of sows with porcine circovirus 2. J Vet Diagn Invest. 1999;11:530-532.
*3. Sorden SD. Update on porcine circovirus and postweaning multisystemic wasting syndrome (PMWS). Swine Health Prod. 2000;8:133-136.
4. Krakowka S, Ellis JA, Meehan BM, Kennedy S, McNeilly F, Allan G. Viral wasting syndrome of swine: experimental reproduction of postweaning multisystemic wasting syndrome in gnotobiotic swine by coinfection with porcine circovirus 2 and porcine parvovirus. Vet Pathol. 2000;37:254-263.
5. Allan GM, Kennedy S, McNeilly F, Foster JC, Ellis JA, Krakowka SJ, Meehan BM, Adair BM. Experimental reproduction of severe wasting disease by co-infection of pigs with porcine circovirus and porcine parvovirus. J Comp Pathol. 1999;121:1-11.
6. Kennedy S, Moffett D, McNeilly F, Meehan BM, Ellis JA, Krakowka S, Allan GM. Reproduction of lesions of postweaning multisystemic wasting syndrome by infection of conventional pigs with porcine circovirus type 2 alone and in combination with porcine parvovirus. J Comp Pathol. 2000;122:9-24.
7. Opriessnig T, Fenaux M, Yu S, Evans RB, Cavanaugh D, Gallup JM, Pallares FJ, Thacker EL, Lager KM, Meng XJ, Halbur PG. Effect of porcine parvovirus vaccination on the development of PMWS in segregated early weaned pigs coinfected with type 2 porcine circovirus and porcine parvovirus. Vet Microbiol. 2004;98:209-220.
8. Harms PA, Sorden SD, Halbur PG, Bolin SR, Lager KM, Morozov I, Paul PS. Experimental reproduction of severe disease in CD/CD pigs concurrently infected with type 2 porcine circovirus and porcine reproductive and respiratory syndrome virus. Vet Pathol. 2001;38:528-539.
9. Rovira A, Balasch M, Segalés J, García L, Plana-Durán J, Rosell C, Ellerbrok H, Mankertz A, Domingo M. Experimental inoculation of conventional pigs with porcine reproductive and respiratory syndrome virus and porcine circovirus 2. J Virol. 2002;76:3232-3239.
10. Allan GM, McNeilly F, Ellis JA, Krakowka S, Meehan BM, McNair I, Walker I, Kennedy S. Experimental infection of colostrum deprived piglets with porcine circovirus 2 (PCV2) and porcine reproductive and respiratory syndrome virus (PRRSV) potentiates PCV2 replication. Arch Virol. 2000;145:2421-2429.
11. Opriessnig T, Thacker EL, Yu S, Fenaux M, Meng XJ, Halbur PG. Experimental reproduction of postweaning multisystemic wasting syndrome in pigs by dual infection with Mycoplasma hyopneumoniae and porcine circovirus type 2. Vet Pathol. 2004;41:624-640.
12. Krakowka S, Ellis JA, McNeilly F, Ringler S, Rings DM, Allan G. Activation of the immune system is the pivotal event in the production of wasting disease in pigs infected with porcine circovirus-2 (PCV2). Vet Pathol. 2001;38:31-42.
13. Albina E, Truong C, Hutet E, Blanchard P, Cariolet R, L'Hospitalier R, Mahe D, Allee C, Morvan H, Amenna N, Le Dimna M, Madec F, Jestin A. An experimental model for post-weaning multisystemic wasting syndrome (PMWS) in growing piglets. J Comp Pathol. 2001;125:292-303.
14. Okuda Y, Ono M, Yazawa S, Shibata I. Experimental reproduction of postweaning multisystemic wasting syndrome in cesarean-derived, colostrum-deprived piglets inoculated with porcine circovirus type 2 (PCV2): investigation of quantitative PCV2 distribution and antibody responses J Vet Diagn Invest. 2003;15:107-114.
15. Bolin SR, Stoffregen WC, Nayar GP, Hamel AL. Postweaning multisystemic wasting syndrome induced after experimental inoculation of cesarean-derived, colostrum-deprived piglets with type 2 porcine circovirus. J Vet Diagn Invest. 2001;13:185-194.
16. Ladekjær-Mikkelsen AS, Nielsen J, Stadejek T, Storgaard T, Krakowka S, Ellis J, McNeilly F, Allan G, Botner A. Reproduction of postweaning multi-systemic wasting syndrome (PMWS) in immuno-stimulated and non-immunostimulated 3-week-old piglets experimentally infected with porcine circovirus type 2 (PCV2). Vet Microbiol. 2002;89:97-114.
17. Resendes A, Segalés J, Balasch M, Calsamiglia M, Sibila M, Ellerbrok H, Mateu E, Plana-Durán J, Mankertz A, Domingo M. Lack of an effect of a commercial vaccine adjuvant on the development of postweaning multisystemic wasting syndrome (PMWS) in porcine circovirus type 2 (PCV2) experimentally infected conventional pigs. Vet Res. 2004;35:83-90.
18. Allan GM, McNeilly F, McNair I, O'Connor M, Meehan, B, Gilpin D, Ellis J, Townsend H, Lasagna C, Boriosi G, Krakowka S. Neonatal vaccination for Mycoplasma hyopneumoniae and post-weaning multisystemic wasting syndrome: a field trial. Pig J. 2001;48:34-41.
19. Kyriakis SC, Saoulidis K, Lekkas S, Militotis ChC, Papoutsis PA, Kennedy S. The effects of immuno-modulation on clinical and pathological expression of postweaning multisystemic wasting syndrome. J Comp Pathol. 2002;126:38-46.
*20. Vanderstichel R, Hurnik D. The effect of a commercial vaccine and adjuvants on the prevalence of post-weaning multisystemic wasting syndrome and related circovirus lesions in a finishing barn. Proc AASV. Orlando, Florida. 2003;34:27-30.
21. Opriessnig T, Yu S, Gallup JM, Evans RB, Fenaux M, Pallares F, Thacker EL, Brockus CW, Ackerman MR, Thomas P, Meng XJ, Halbur PG. Effect of vaccination with selective bacterins on conventional pigs infected with type 2 porcine circovirus. Vet Pathol. 2003;40:521-529.
22. Allan GM, McNeilly F, McNair I, Kennedy S, Meehan B, Ellis J, Krakowka S. Immunostimulation, PCV-2 and PMWS. Vet Rec. 2000;170-171.
23. Nawagitgul P, Harms PA, Morozov I, Thacker BJ, Sorden SD, Lekcharoensuk C, Paul PS. Modified indirect porcine circovirus (PCV) type 2-based and recombinant capsid protein (ORF2)-based enzyme-linked immunosorbent assays for detection of antibodies to PCV. Clin Diagn Lab Immunol. 2002;9:33-40.
24. Fenaux M, Halbur PG, Gill M, Toth TE, Meng XJ. Genetic characterization of type 2 porcine circovirus (PCV-2) from pigs with postweaning multisystemic wasting syndrome in different geographic regions of North America and development of a differential PCR-restriction fragment length polymorphism assay to detect and differentiate between infections with PCV-1 and PCV-2. J Clin Microbiol. 2000;38:2494-2503.
25. Fenaux M, Halbur PG, Haqshenas G, Royer R, Thomas P, Nawagitgul P, Gill M, Toth TE, Meng XJ. Cloned genomic DNA of type 2 porcine circovirus is infectious when injected into the liver and lymph nodes of pigs: characterization of clinical disease, virus distribution, and pathologic lesions. J Virol. 2002;76:541-551.
26. Halbur PG, Paul PS, Frey ML, Landgraf J, Eernisse K, Meng XJ, Lum MA, Andrews JJ, Rathje JA. Comparison of the pathogenicity of two U. S. porcine reproductive and respiratory syndrome virus isolates with that of the Lelystad virus. Vet Pathol. 1995;32:648-660.
27. Sorden SD, Harms PA, Nawagitgul P, Cavanaugh D, Paul PS. Development of a polyclonal-antibody-based immunohistochemical method for the detection of type 2 porcine circovirus in formalin-fixed, paraffin-embedded tissue. J Vet Diagn Invest. 1999;11:528-530.
28. Vicca J, Stakenborg T, Maes D, Butaye P, Peeters J, de Kruif A, Haesebrouck F. Evaluation of virulence of Mycoplasma hyopneumoniae field isolates. Vet Microbiol. 2003;97:177-190.
* Non-refereed references.;