Original research |
Peer reviewed |
Palpable lingual cysts, a possible indicator of porcine cysticercosis, in Teso District, Western Kenya
Incidencia de quistes linguales palpables, un posible indicador de la cisticercosis en el Distrito de Teso, Kenya del Oeste
Occurrence de kystes linguaux palpables, un indicateur possible de cysticercose porcine dans le district de Teso, Kenya Occidental
Florence K. Mutua, BVM, MSc; Thomas F. Randolph, PhD; Samuel M. Arimi, BVM, MSc, PhD; Philiph M. Kitala, BVM, MSc, PhD; Samuel M. Githigia, BVM, MSc, PhD; Arve L. Willingham, DVM, MSc, PhD; Francis M. Njeruh, BVM, MSc, PhD
FKM, SMA, PMK, FMN: Department of Public Health, Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Nairobi, Nairobi, Kenya. TFR: International Livestock Research Institute, Nairobi, Kenya. SMG: Department of Pathology, Microbiology and Parasitology, Faculty of Veterinary Medicine, University of Nairobi, Nairobi, Kenya. ALW: WHO/FAO Collaborating Center for Parasitic Zoonoses, Royal Veterinary and Agricultural University, Frederiksberg, Denmark. Corresponding author: Dr Florence K. Mutua, Department of Public Health, Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Nairobi, PO Box 29053, Nairobi, Kenya; Tel: +254-07-33-546859; E-mail: flmutua@yahoo.com.
Cite as: Mutua FK, Randolph TF, Arimi SM, et al. Palpable lingual cysts, a possible indicator of porcine cysticercosis, in Teso District, Western Kenya. J Swine Health Prod. 2007;15(4):206–212.
Also available as a PDF.
SummaryObjectives: To estimate the prevalence of palpable lingual cysts in pigs in Western Kenya, a possible indicator of porcine cysticercosis, and to study the potential risk factors associated with this clinical finding. Methods: During a cross-sectional survey, 316 randomly selected small-scale farms were visited, from which a case-control study of 31 case farms and 93 randomly selected control farms was constructed. Information on potential risk factors for Taenia solium cysticercosis-taeniosis was obtained using questionnaires administered via personal interviews. Results: Farm prevalence of palpable lingual cysts was estimated at 9.8% (31 of 316) (95% confidence interval [CI], 6.5%-13.1%). Total number of pigs testing positive was 33, resulting in a pig prevalence of 6.5% (95% CI, 4%-9%). Pigs were kept as a source of income (98%) and for home consumption (2%). Sources of pigs included local purchases (94%; 117 of 124), and purchases from Uganda (6%; seven of 124). Most farmers (95%; 118 of 124) kept their pigs on free range. Pork was sourced from local butcheries (85%) and home slaughtering (15%). Most households slaughtering pigs at home had their pork “inspected” by household friends (five of nine). Absence of latrines was more common in case households (42%; 13 of 31) than in controls (18%; 17 of 93) (P = .01; OR = 3.2; 95% CI, 1.2%-8.55). Implications: Palpable lingual cysts are prevalent in the locally raised pigs of Western Kenya. Further studies using more sensitive diagnostic tests are required to confirm the risk of porcine cysticercosis. | ResumenObjetivos: Calcular la prevalencia en cerdos en Kenya del Oeste de quistes linguales palpables, un posible indicador de la cisticercosis porcina, y estudiar los factores de riesgo potenciales asociados con este hallazgo clínico. Métodos: Durante una investigación transversal, se visitaron 316 granjas de pequeña producción seleccionadas al azar, con las cuales se estableció un estudio de caso-control de 31 granjas caso y 93 granjas control seleccionadas al azar. Se obtuvo información de factores de riesgo potenciales para la Taenia solium cisticercosis-taeniosis utilizando cuestionarios vía entrevistas personales. Resultados: La prevalencia en granja de quistes linguales palpables se calculó en 9.8% (31 de 316) (95% intervalo de confianza [CI], 6.5%-13.1%). El número total de cerdos positivos fue de 33, resultando en una prevalencia de 6.5% (95% CI, 4%-9%). Los cerdos se utilizaban como una fuente de ingreso (98%) y para consumo casero (2%). Las fuentes de cerdos incluyeron compras locales (94%; 117 de 124), y compras en Uganda (6%; siete de 124). La mayoría de los productores (95%; 118 de 124) mantuvieron sus cerdos en pastoreo. El cerdo provenía de las carnicerías locales (85%) y rastros caseros (15%). La mayoría de cerdos sacrificados en casa pidieron a los amigos de la familia (cinco de nueve) “inspeccionar” a sus cerdos. La ausencia de letrinas fue más común en los casos caseros (42%; 13 de 31) que en los controles (18%; 17 de 93) (P = .01; OR = 3.2; 95% CI, 1.2%-8.55). Implicaciones: Los quistes linguales palpables son prevalentes en los cerdos criados localmente de Kenya del Oeste. Se requiere de estudios adicionales que utilicen pruebas diagnósticas más sensibles que confirmen el riesgo de cisticercosis porcina. | ResuméObjectifs: Estimer la prévalence de kystes linguaux palpables chez des porcs du Kenya Occidental comme indicateur possible de cysticercose porcine et étudier les facteurs de risque potentiels associés à ces trouvailles cliniques. Méthodes: Au cours d’une étude transversale, 316 fermes de petite envergure ont été sélectionnées au hasard et visitées. De ce groupe, une étude de cas-témoins a été élaborée utilisant 31 fermes-cas et 93 fermes-témoins sélectionnées au hasard. Des informations sur les facteurs de risque potentiels pour la cysticercose-taeniose à Taenia solium ont été obtenues à l’aide d’un questionnaire administré lors d’entrevue personnelle. Résultats: La prévalence de fermes o Implications: Des kystes linguaux palpables sont prévalents chez les porcs élevés localement au Kenya Occidental. Des études supplémentaires utilisant des méthodes diagnostiques plus sensibles sont nécessaires pour confirmer les risques de cysticercose porcine. |
Keywords: swine, Taenia
solium, cysticercosis, taeniosis, lingual cysts
Search the AASV web site
for pages with similar keywords.
Received: July
6, 2006
Accepted: February
7, 2007
Taenia solium taeniosis-cysticercosis remains a major public health problem in many developing countries of Latin America, Africa, and Asia.1 The life cycle includes the pig as the intermediate host, harboring larval cysts (Cysticercus cellulosae), and humans as the definitive hosts, harboring adult tapeworms (taeniosis). Humans can also serve as intermediate hosts if they consume T solium eggs. Neurocysticercosis, which is infection of the human brain by the larval stage of the parasite, is reportedly the major cause of preventable epilepsy in developing countries.2 However, the true impact of the disease has been obscured by unavailability of the sensitive and specific diagnostic tools necessary for collection of reliable epidemiological data, eg, the antigen ELISA (Ag Elisa; University of Zambia, Lusaka, Zambia).3,4 In Kenya, very little epidemiological work has been done on taeniosis-cysticercosis, and it is assumed to be rare.5 Few cases have been reported in pigs during routine meat inspection. The lingual examination method has been used to detect palpable cysts, which may indicate porcine cysticercosis. Use of the lingual palpation method to detect porcine cysticercosis has been questioned. This method requires technical expertise,6 is of low sensitivity, and is capable of detecting C cellulosae cysts only in heavily infected pigs.2,7,8 This method has been used, however, because it is readily available and is less costly than Ag ELISA testing. Githigia et al5 examined a total of 293 pigs in the South Nyanza and Busia districts of Kenya using lingual palpation and reported a prevalence rate of 10% to 14%. The objectives of this study were to estimate the prevalence of palpable lingual cysts in pigs in Teso District and to determine the potential risk factors for occurrence of this clinical sign. The information thus gained will be essential for better understanding of the porcine cysticercosis situation in Western Kenya and in planning informed control strategies for combating the disease in this area.
Materials and methods
Study area and household sampling
This study was conducted between September 2003 and May 2004 in Teso District, Western Kenya. This district lies between latitudes 0� 29´ and 0� 32´ north and longitudes 34� 01´ and 34� 07´ east and has an area of 559 km2. Most parts of the district receive between 1270 and 1790 mm mean annual rainfall, suitable for both food and cash crops. The pig population in Teso District, according to government annual reports of 1995 to 2000,9 were as follows: 1995, 2590; 1996, 2500; 1997, 2100; 1998, 2610; 1999, 2700; and 2000,1493. The pig-human ratio, calculated as the proportion of pig farmers divided by the total number of households in the district, was 1:14, a point estimate for the year 1999. Pigs reared are mainly of the local breed, and the majority are kept under free-range conditions. Other livestock kept include cattle, sheep, goats, donkeys, rabbits, and poultry.5,10
The population of interest in the present study consisted of pigs kept by small-scale pig farmers in Teso District. Small-scale pig keepers are families who own one to three pigs, on average. Each district in Kenya is divided into divisions and these are divided into locations. Two locations in each of the four divisions of the district were selected for convenience, ie, if there was road access to the locations as well as a known history of small-scale pig farming. Subsequently, a list of all small-scale pig farmers in each of the selected locations was obtained through the assistance of the local administration, including village headmen, assistant chiefs, and chiefs. Out of the resultant household sampling frame of 540 households, a cross-sectional sample of 316 households was randomly selected using a random-number table. Randomization was based on farms within the locations. Farmers who had sold their pigs were replaced by initially nonselected study households drawn randomly from the same list.
Estimation of prevalence of palpable lingual cysts
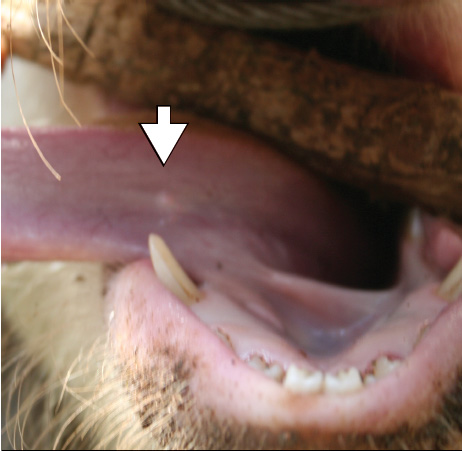
Pigs in the 316 households were examined for C cellulosae using the lingual palpation method.5,7,11 Briefly, pigs were snared, a walking stick was inserted horizontally into the mouth to act as gag, and then the pig’s tongue was extended by holding it with a cotton cloth to enable palpation. A cyst was defined as any palpable whitish vesicular mass, measuring approximately 1 to 2 mm, on the underside of the tongue (Figure 1). Use of animals in this study was approved by the Kenya Ministry of Livestock and Fisheries.
Figure 1: Lingual palpation method for detecting larval cysts of Taenia solium in pigs. Arrow identifies a lingual cyst.
|
Determination of risk factors for palpable cysts indicative of porcine cysticercosis
In order to determine the risk factors for palpable lingual cysts in the district, a case-control sampling strategy was conducted within the randomly selected 316 households. A case household was one that had at least one pig testing positive by the lingual palpation method. A control household was one in which no pig tested positive using this method. For every case household identified, three control households were randomly selected, according to Dohoo et al.12 All cases were included in this portion of the study. The cases and the randomly identified control households were then visited and questionnaires (Figure 2) were administered via personal interviews. An animal-health worker, blinded to the disease status of the household, assisted in the administration of the questionnaire using the local Teso language. A total of 124 households, comprising 31 cases and 93 controls, were visited. The questionnaire sought information on pig management practices, sources of pigs, knowledge of T solium taeniosis-cysticercosis, and hygiene practices.
Figure 2: Survey questionnaire concerning porcine cysticercosis administered to 124 randomly identified households that owned one to three pigs in Teso District, Western Kenya. Personal interviews were conducted between September 2003 and May 2004. (Please refer to the
|
Data entry and analysis
Data were entered into Microsoft Excel (Microsoft Corp, Reading, UK) and exported to GenStat 7th edition (VSN International Ltd, Hemel Hempstead, UK) for statistical analysis. The farm prevalence of palpable lingual cysts was computed by dividing the number of farms that had at least one pig that tested positive (numerator) by the total number of farms visited (denominator). Prevalence in pigs was calculated as a proportion of the total number of palpable lingual cysts (numerator) divided by the total number of pigs examined (denominator).12,13 Associations between the potential risk factors for lingual palpable cysts in pigs were assessed using the chi-square statistic and Fisher’s exact test, and the strengths of the associations were determined using the odds ratio (OR). Risk factors with P < .05 were considered significant.13,14 We also calculated the estimated population attributable fraction (PAF) and the estimated attributable fraction (AF) for the risk factor “farm without a latrine.” The PAF was calculated using the following equation: estimated PAF = OR – (1 ÷ OR). The PAF is the contribution of lack of latrines to occurrence of cysticercosis in the whole population of pigs. In this case, PAF is a function of both the frequency of households having no latrine and the strength of the association between lack of a latrine and cysticercosis. The AF is the proportion of pigs with palpable lingual cysts that was due to the risk factor “farm without a latrine.”13 The AF was calculated using the following equation: estimated AF = 1 – [( c � (b + d)) ÷ (d � (a + c))], where a, b, c, and d represent possible disease and exposure factor status: a, positive pigs from farms without latrines; b, negative pigs from farms without latrines; c, positive pigs from farms with latrines; d, negative pigs from farms with latrines.
Results
Prevalence of palpable lingual cysts
A total of 505 pigs from the cross-sectional sample of 316 study households were examined. Of these, 5% were substitute farms, ie, they replaced non-pig-keeping farms that had initially been selected for the study. The farm prevalence of palpable lingual cysts was estimated at 9.8% (31 of 316) (95% CI, 6.5%-13.1%). The total number of pigs testing positive was 33, resulting in a pig prevalence of 6.5% (33 of 505) (95% CI, 4.0%-9.0%). Most positive pigs (91%; 30 of 33) had one cyst detected on lingual examination. Most farmers (94%; 297 of 316) reported that pig diseases were a common health problem affecting pig-keeping in the area. None of the farmers interviewed had any knowledge of cysticercosis in pigs.
Pig husbandry practices
Pigs in the district were kept for income (98%) and home consumption (2%). Major sources of pigs were purchases (98%; 122 of 124) and gifts from friends (2%; 2 of 124). Few households had purchased pigs from Uganda (6%; seven of 124), and only one of these seven was a case household. A total of six households (5%) confined their pigs by tethering (four of six) and housing (two of six). One of the two households that reportedly housed their pigs was a case household. Ninety-five percent of pig farmers (118 of 124) allowed their pigs to roam freely in the villages, and 71% of these farmers (84 of 118) reported combining tethering and free ranging. Tethering was mostly practiced during rainy seasons, to prevent scavenging pigs from destroying crops in the local farms. Pigs allowed to scavenge were identified by farmers interviewed as major causes of neighborhood conflicts in the study area. Free ranging was reported to be very economical, since pigs could easily scavenge for feeds, incurring little or no maintenance cost.
Public awareness of porcine cysticercosis and human taeniosis
Very few respondents (16%; 19 of 120) knew that tapeworm infestation in humans occurred because of consumption of inadequately cooked meat. Some thought that tapeworms occurred because of consumption of dirty foods (10%; 12 of 120), while others attributed it to consumption of raw cassava (27%; 33 of 120), a root crop that is a staple in the area. The majority of the households (76%; 94 of 124) had latrines. Latrines were absent in 42% and 18% of the case and control households, respectively (Table 1). Home garbage disposal was either in the nearby crop-growing fields (71%) or in a household garbage pit (29%). Thirty-two percent (10 of 31) of the case households and 28% (26 of 93) of the control households had garbage pits. All home garbage pits were shallow and were not fenced off, and pigs could easily access the garbage. Home slaughter of pigs was reported in 15% of the 110 pork-consuming households. Of the nine “home-inspected” households, 56% (five of nine) were “inspected” by household friends.
Table 1: Comparison of case and control households in Teso District, Kenya, by suspected risk factors for porcine cysticercosis-taeniosis*
* Information on potential risk factors for Taenia solium cysticercosis-taeniosis was obtained using questionnaires administered via personal interviews conducted between September 2003 and May 2004. † Kenya-Uganda border. ‡ Unrestricted pigs were able to forage free range; restricted pigs were either housed or tethered. |
Absence of latrines was significantly higher (P = .01) in the case households than in the control households, and households without latrines were three times more likely than households with latrines to have pigs with palpable lingual cysts (Table 1). The estimated AF was 69%, indicating that in 69% of cases, in pigs from households without latrines, the presence of palpable lingual cysts was associated with the absence of latrines. The estimated PAF was 29%, indicating that in this population, the provision of latrines in each household would be expected to reduce the prevalence of lingual cysts by 29%. A total of 70 of 124 respondents reported occurrence of epilepsy in their villages, and 24 (19%) had observed epileptics in their families. These proportions did not differ by case status.
Discussion
Emergence of T solium cysticercosis and taeniosis has been identified as one of the major constraints to profitable pig production in the developing world. It is a serious public health risk in Latin America, Asia, and Africa.1,15,16 There exists very little baseline data on the occurrence of porcine cysticercois in Kenya.5,17 Cases of cysticercosis have been identified during routine meat inspection in a major pig abattoir in the country.17 There are no published reports on the occurrence of T solium metacestodes in Teso District. This study serves to confirm the presence of palpable lingual cysts in the area. Although no data on the profitability of small-scale pig farming is available in Kenya, this study showed that small-scale pig keeping contributes to the livelihood of the local Teso people. Previous reports have identified pig farming as more profitable than other types of livestock keeping, possibly because it generates greater incomes to farmers.18
Porcine cysticercosis was not identified by farmers as a common problem affecting small-scale pig farming. Cysticercosis in pigs is subclinical. Unless pigs are slaughtered and infection pointed out by meat inspectors, farmers may be unaware of the infection. Examination of pigs for palpable cysts of T solium and alerting the farmers of the legislative and public health implications may reduce the possible losses that could arise due to carcass condemnation during postmortem meat inspection.7,19 However, Rodriquez-Hildago et al19 observed that pigs with palpable cysticerci were not presented for official slaughter, and were instead slaughtered privately.
Results of this study indicate that palpable lingual cysts are prevalent in the locally raised pigs of Teso District. The prevalence of palpable cysts observed in this study (6.5%) falls within the estimated range of 5% to 30% reported in most endemic areas elsewhere in the world.20 In addition, this compares with the 5.5% prevalence obtained in a similar study conducted in Nigeria by Onah and Chiejina.21 In the Republic of Tanzania, a prevalence of up to 17% has been reported in Mbulu District using the lingual palpation method.7,11,22 Zoli et al4 reported prevalences of 26% and 12%, based on postmortem results, in Chad and Ghana, respectively. In a recent study conducted in the neighbouring Busia and Nyanza Districts of Kenya, a prevalence of porcine cysticercosis between 10% and 14% was reported.5
The sensitivity of the lingual palpation method in detecting porcine cysticercosis has previously been questioned.8,23 This method requires technical expertise, is of low sensitivity, and is capable of detecting C cellulosae cysts only in heavily infected pigs. Gonzalez et al23 reported a sensitivity of 70% and a specificity of 100% in a study conducted in Peru. In an abattoir survey conducted in Zambia, the relative sensitivity of tongue palpation, compared to visual inspection of the carcass, was only 43%.8 It is expected that the 6.5% pig-level prevalence in Teso district is an underestimate of the true prevalence, which may be as high as 9.3% to 15%.
Several cases of porcine cysticercosis were detected in a major abattoir in Nairobi in 1988;17 the pigs had reportedly been imported from the Republic of Tanzania.However, of the 31 case households considered in this study, 30 purchased their pigs from within Kenya, indicating that control strategies need to be focused on the locally raised pigs.
Previous studies have reported a significant association between having a farm latrine and occurrence of porcine cysticercosis.7 Other potential risk factors that could be associated with porcine cysticercosis, such as history of tapeworm carriers in the families, were surprisingly insignificant in this study. The lack of association of these other risk factors and palpable cysts indicative of porcine cysticercosis may be due to the uniform nature of the risk factors across the population examined. For example, most farmers surveyed (95%) allowed their pigs to roam freely in the villages. This is, however, against the Kenyan laws.24 As it is believed that housing reduces the risk of cysticercosis, it is interesting to note that one of the two households that housed their pigs was a case household. All pigs had access to household garbage. It is probable that the sample size used was small and the study lacked the power to detect a difference.12 The limited sensitivity of the lingual test may also have contributed to the lack of association between these explanatory variables and the outcome of interest.
Case-control studies are characterized by a number of biases.13 Ensuring that the person administering the questionnaire was blinded to the case status of the farmer eliminated one source of potential bias. However, due to the low sensitivity of the lingual palpation method, it was not possible to ascertain whether the controls were truly disease free, and therefore, false-negatives cannot be ruled out. This would have biased the factor analysis towards the null hypothesis, reducing the ability to identify factors associated with the disease. In the future, the use of the parasite antigen-detecting ELISA, which has a higher sensitivity and specificity, should be used to discriminate cases and controls.
Implications
- Palpable cysts indicative of porcine cysticercosis are common in the locally raised pigs of Teso District.
- There is a need to employ more sensitive serological tests to confirm the findings of this study.
- There is a need to formulate and implement appropriate control measures for this serious zoonosis in Teso District.
- The current low levels of awareness suggest that educational programs should be targeted at the general public (including farmers) as well as public health and medical personnel.
Acknowledgements
This study was funded by the International Livestock Research Institute (ILRI). We thank the DANIDA-funded ENRECA Livestock Helminths Research Project in Eastern and Southern Africa and the Faculty of Veterinary Medicine, University of Nairobi, for their support throughout the study period. We thank Esther Schelling (ILRI) for her inputs into the manuscript. We appreciate the logistic and technical support given by the Ministry of Livestock and Fisheries Development, Ministry of Health, Teso District Hospital, and the provincial administration in the course of this study. The small-scale pig keepers in Teso District are thanked for their cooperation.
References
1. Sarti E, Schantz PM, Plancarte A, Wilson M, Gutierrez O, Lopez A, Roberts J, Flisser A. Prevalence and risk factors for Taenia solium taeniosis/cysticercosis in humans and pigs in a village in Morelos, Mexico. Am J Trop Med Hyg. 1992;46:677–684.
2. Garcia H, Pretell EJ, Gilman RH, Martinez SM, Moulton LH, Del Brutto OH, Herrera G, Evan CA, Gonzales AE. A trial of antiparasitic treatment to reduce the rate of seizures due to cerebral cysticercosis. N Eng J Med. 2004;350:249–258.
3. Tsang VC, Wilson M. Taenia solium cysticercosis. An under-recognized but serious public health problem. Parasitol Today. 1995;11:124–126.
4. Zoli A, Oliver S, Assana E, Pierre J, Dorney P, Brandt J, Geerts S. Regional status, epidemiology and impact of Taenia solium cysticercosis in Western and Central Africa. Acta Trop. 2003;87:35–42.
*5. Githigia SM, Murekefu K, Ngesa SM, Otieno RO, Kahai R. The prevalence of porcine cysticercosis and risk factors in Busia District, Kenya. Proc 11th Ann Meet ENRECA Livest Res Proj Eastern Southern Africa. Lusaka, Zambia. 2002:30.
6. Sciutto E, Martinez J, Villalobos N, Hernandex M, Jose M, Beltran C, Rodarte F, Flores I, Bobadilla JR, Fragosso G, Parhhouse M, Harrison L, de Aluja A. Limitations of current diagnostic procedure for diagnosis of Taenia solium cysticercosis in rural pigs. Vet Parasitol. 1998;79:299–313.
7. Ngowi H, Kassuku A, Maeda G, Boa M, Carabin H, Willingham AL. Risk factors for prevalence of porcine cysticercosis in Mbulu District, Tanzania. Vet Parasitol. 2004;120:275–283.
8. Phiri IK, Dorney P, Gabriel S, Willingham A, Speybroeck N, Vercruysse J. The prevalence of porcine cysticercosis in Eastern and Southern Provinces of Zambia. Vet Parasitol. 2002;108:31–39.
9. Ministry of Agriculture, Livestock Development and Marketing 1986–2000. Republic of Kenya, Department of Livestock Production, Animal Production Division. Annual Reports.
10. Government of Kenya, Ministry of Finance and Planning. Effective management for sustainable economic growth and poverty reduction. Teso District Development Plan (2002–2008).
11. Phiri IK, Ngowi H, Afonso S, Matega E, Boa M, Mukaratirwa S, Githigia SM, Saimo MK, Sikasunge CS, Maingi N, Lubega GW, Kassuku A, Michael LM, Siziya S, Krecek RC, Noormahomed E, Vilhena M, Dorney P, Willingham AL. The emergence of Taenia solium cysticercosis in Eastern and Southern Africa as a serious agricultural problem and public health risk. Acta Tropica. 2003;87:25–33.
12. Dohoo I, Martin W, Stryhn H. Veterinary Epidemiologic Research. University of Prince Edward Island, Charlottetown, Prince Edward Island, Canada: AVC Publishers; 2003:163–174, 335–386.
13. Martin S, Allan H, Preben W. Veterinary Epidemiology: Principles and Methods. Ames, Iowa: Iowa State University Press; 1987:32–45.
14. Hosmer DW, Lemeshow S. Applied Logistic Regression. Hoboken, New Jersey: John Wiley and Sons Inc; 1989:82–133.
*15. Ruiz A. Prevalence and control of taeniosis and cysticercosis, a global perspective. Proc Int Workshop Cysticercosis. Onderstepoort, South Africa. 1997;1–18.
16. Mafojane NA, Appleton C, Krecek RC, Michael LM, Willingham AL. The status of neurocysticercosis in Eastern and Southern Africa. Acta Tropica. 2003;87:25–33.
*17. Otwelo J. An overview of Cysticercus cellulosae in Kenya. A presentation during a cysticercosis workshop in Arusha, Tanzania. April 19–22, 2002.
18. Lekule F, Kyvsgaard N. Improving pig husbandry in tropical resource-poor communities. Acta Trop. 2003;87:111–117.
19. Rodriguez-Hildago R, Benitez-Ortiz W, Dorney P, Geerts S, Geysen D, Ron-Roman J, Proana-Parez F, Chavez-Larrea MA, Barrionueva-Samaniego M, Celi-Erazo M, Vizcaino-Ordonez L, Brandt J. Taeniosis/cysticercosis in man and in animals in the Sierra of Northern Ecuador. Vet Parasitol. 2003;118:51–60.
20. Craig P, Rogan MT, Allan JC. Detection, screening and community epidemiology of taeniid cestode zoonoses: cystic echinococcosis, alveolar echinococcosis and neurocysticercosis. Adv Parasitol. 1996;38:169–250.
21. Onah DN, Chiejina SN. Taenia solium cysticercosis and human taeniasis in the Nsukabarea of Enugu State, Nigeria. Ann Trop Med Parasitol. 1995;89:399–407.
22. Boa ME, Bogh HO, Kassuku AA, Nanzen P. The prevalence of T. solium metacestodes in pigs in northern Tanzania. J Helminthol. 1995;67:113–117.
23. Gonzalez AE, Cama V, Gilman RH, Tsang VC, Pitcher JB, Chevera A, Castro M, Montenegro T, Verastegui M, Miranda E. Prevalence and comparison of serological assays, necropsy, and tongue examination for the diagnosis of porcine cysticercosis in Peru. Am J Trop Med Hyg. 1990;43:194–199.
24. Government of Kenya. The Animal Diseases Act, 1972. Chapter 364. Revised Edition. Printed and published by the Government Printer.
* Non-refereed references.