Case report |
Peer reviewed |
Use of a one-dose subunit vaccine to prevent losses associated with porcine circovirus type 2
Uso de una de vacuna de subunidades de una dosis para prevenir pérdidas asociadas con el circovirus porcino tipo 2
Utilisation d’un vaccin sous-unitaire à dose unique pour prévenir les pertes associées au circovirus porcin de type 2
Robert Desrosiers, DVM, Dipl ABVP; Edward Clark, DVM, MVSc, Dipl ACVP; Dorine Tremblay, DVM; Robert Tremblay, DVM, DVSc, Dipl ACVIM; Dale Polson, DVM, PhD
RD, RT: Boehringer Ingelheim Canada, Burlington, Ontario, Canada. EC: CARE Centre, Calgary, Alberta, Canada. DT: Vetcoporc Consultants, St-Lambert-de-Lauzon, Québec, Canada. DP: Boehringer Ingelheim Vetmedica Inc, Ames, Iowa. Drs Robert Desrosiers and Robert Tremblay were employed by Boehringer Ingelheim Canada and Dr Dale Polson was employed by Boehringer Ingelheim Vetmedica Inc at the time this study was conducted. Corresponding author: Dr Robert Desrosiers, 2235 Carillon, Saint-Hyacinthe, Québec, Canada J2S 7W1; Tel: 450-773-8929; Fax: 450-773-8932.
Cite as: Desrosiers R, Clark E, Tremblay D, et al. Use of a one-dose subunit vaccine to prevent losses associated with porcine circovirus type 2. J Swine Health Prod. 2009;17(3):148–154.
Also available as a PDF.
SummaryIn a commercial swine herd in eastern Canada, porcine circovirus associated disease (PCVAD) had been occurring for about 18 months when a test vaccine was introduced. Pigs were affected approximately 3 weeks after entering the finishing units. A total of 3852 pigs were enrolled in the study. Half of the pigs were randomly selected to be vaccinated at 19 to 59 days of age with a one-dose porcine circovirus type 2 (PCV2) vaccine (Ingelvac CircoFlex; Boehringer Ingelheim Vetmedica Inc, St Joseph, Missouri). The other half of the pigs were injected on the same day with sterile water (controls). No local or systemic adverse reactions were observed. The study pigs were placed in four different finishing units and in each one, mortality was significantly lower in vaccinated pigs (P < .01). Enlarged lymph nodes and white spots on the kidneys were among the most frequent lesions suggesting PCVAD. Splenic infarcts were observed in a few pigs. Results of two different serological tests showed that pigs could be effectively vaccinated in the presence of maternal immunity. There was no correlation between seroconversion (in response to vaccination) and protection. | ResumenEn un hato porcino comercial del este de Canadá, la enfermedad asociada al circovirus porcino (PCVAD por sus siglas en inglés) estuvo presente durante los 18 meses anteriores al momento en el que se introdujo una vacuna de prueba. Los cerdos se afectaban aproximadamente 3 semanas después de entrar a las unidades de finalización. Un total de 3852 cerdos participaron en el estudio. Se seleccionó al azar a la mitad de los cerdos para vacunarlos entre los 19 y 59 días de edad con una vacuna de una dosis (Ingelvac CircoFlex; Boehringer Ingelheim Canadá, Burlington, Ontario, Canadá) contra circovirus porcino tipo 2 (PCV2 por sus siglas en inglés). La otra mitad de los cerdos se inyectó el mismo día con agua estéril (controles). No se observaron reacciones adversas sistémicas ó locales. Los cerdos del estudio se colocaron en cuatro unidades de finalización diferentes y en cada una, la mortalidad fue significativamente más baja en los cerdos vacunados (P < .05). Entre las lesiones más frecuentes se encontraron nódulos linfáticos aumentados de tamaño y manchas blancas en riñones sugiriendo la presencia de PCVAD. Se observaron infartos en el bazo en pocos cerdos. Los resultados de dos pruebas serológicas diferentes mostraron que los cerdos podían ser vacunados eficazmente frente a la presencia de inmunidad materna. No hubo correlación entre la seroconversión (respuesta a la vacunación) y la protección. | ResuméDans un troupeau porcin commercial de l’est du Canada, les maladies associées au circovirus porcin (PCVAD) étaient rencontrées depuis environ 18 mois lorsqu’un vaccin à l’essai a été introduit. Les porcs étaient affectés environ 3 semaines après leur entrée dans les unités de finition. Au total, 3852 porcs ont été inclus dans cette étude. La moitié des porcs ont été sélectionnés au hasard pour être vaccinés entre 19 et 59 jours d’âge avec un vaccin à dose unique dirigé contre le circovirus porcin de type 2 (PCV2) (Ingelvac CircoFlex; Boehringer Ingelheim Canada, Burlington, Ontario, Canada). L’autre moitié des porcs a été injectée au même jour avec de l’eau stérile (témoins). Aucune réaction adverse locale ou systémique n’a été observée. Les porcs à l’étude ont été mis dans quatre unités de finition différentes et dans chacune la mortalité était significativement moindre chez les animaux vaccinés (P < .05). Une hypertrophie des ganglions lymphatiques et des zones blanchâtres sur les reins étaient parmi les lésions les plus fréquentes suggérant le PCVAD. Des infarcti spléniques ont été observés chez quelques porcs. Les résultats de deux épreuves sérologiques différentes ont démontré que les porcs pouvaient être vaccinés de manière efficace en présence d’immunité maternelle. Il n’y avait aucune corrélation entre séroconversion (en réponse à la vaccination) et protection. |
Keywords: swine, porcine circovirus associated disease,
porcine circovirus vaccine, mortality, serological response, PCV2, PCVAD
Search the AASV web site
for pages with similar keywords.
Received: February 1, 2008
Accepted: June 12, 2008
Porcine circovirus type 2 (PCV2) is considered an essential element for the development of porcine circovirus associated disease (PCVAD), formerly designated as postweaning multisystemic wasting syndrome.1 This paper describes results obtained with a one-dose subunit PCV2 vaccine (Ingelvac CircoFlex; Boehringer Ingelheim Vetmedica Inc, St Joseph, Missouri) to prevent clinical signs associated with PCVAD in pigs in a commercial swine herd vaccinated at different ages during the epizootic that occurred in Canada in 2005 and 2006. Control and vaccinated pigs were compared in terms of mortality, pathological lesions, and serological results.
Case description
Production system
The system chosen for the study consisted of a 1300-sow herd in eastern Canada, with an offsite nursery and four finishing units all on separate sites. The system was negative for porcine reproductive and respiratory syndrome virus and Mycoplasma hyopneumoniae on the basis of history, absence of clinical signs, minimal lung lesions at slaughter, and serological results. The system had suffered PCVAD losses for about 18 months before the study began, with mortality rates approximately 6% to 12% during the outbreak, increasing from approximately 1.5% to 4% before the outbreak started. Losses occurred exclusively in finishing units, usually beginning 3 to 4 weeks post placement, and were confirmed to be associated with PCVAD by the presence of classical histological lesions and demonstration of the organism in affected tissues. The performance of the sows (27 pigs per sow per year) and nursery pigs (approximately 1% mortality) was excellent and was not affected by the outbreak of PCVAD in the finishing units. The sow herd was not vaccinated against PCV2. The system was managed to respect the Canadian recommended code of practice for the care and handling of pigs.2
Vaccination and pen allocation
An independent vaccination crew was hired to inject the pigs so that the entire staff of the farms involved and the clinical investigators (RD and DT) would be blinded to vaccine and control pens. The nursery site comprised four identical buildings, each with four rooms of approximately 275 pigs distributed among 12 pens. At arrival, the study pigs were placed in pens according to their size. In each room, the toss of a coin determined whether the first pen received product A or product B, and then pens alternated by treatment so that potential exposure to microorganisms and environmental stresses would be similar for both groups. The two groups comprised pigs that appeared to be approximately the same size.
On the day of injection, the vaccination crew evaluated the study pigs for post-injection reactions (eg, apathy, swelling, death). During the next 3 days, one investigator (RD) evaluated local and systemic reactions in the study pigs. Afterwards, one investigator (RD or DT) made weekly visits to evaluate both the safety and efficacy of the tested products. Safety was assessed by clinically evaluating the pigs for signs of adverse reactions in the hours and days following vaccination. Lack of efficacy was determined on the basis of pigs showing clinical signs and lesions of PCVAD.
The two treatment groups received by intramuscular injection either 1 mL of sterile water (Eau Stérile Injection USP; Vétoquinol N-A Inc, Lavaltrie, Québec, Canada; 1933 controls) or 1 mL of the Ingelvac CircoFlex vaccine (1919 vaccinates). Injections were administered on a single day; thus, pigs between 19 and 59 days of age were injected.
As pigs normally left the nursery at an average of 62 days of age, those injected at 59 days of age were introduced into the first finishing unit only 3 days after treatment. The first finishing barn was filled with pigs that had been injected at 45 to 59 days of age (Barn 1). The other three barns were filled with pigs that had been injected at 38 to 45 days of age (Barn 2), 22 to 36 days of age (Barn 3), and 19 to 22 days of age (Barn 4). In all four finisher barns, pens of group A pigs alternated with pens of group B pigs. The first pen in each barn, as the result of a coin toss, received pigs from group A and then pens for treatment groups alternated as in the nurseries.
Investigators’ assessments
The manager of the four finishing units included in the trial waited until the investigator arrived for the weekly visit before euthanizing pigs so that animals showing clinical signs representative of PCVAD could be necropsied. Pigs to be euthanized were selected on the agreement of the investigator and manager of the farm, taking into consideration the likelihood that the animals would live, the suffering endured, and the probability that they would be condemned at the abattoir if kept alive. Pigs that died within a day of the investigator’s visit, and in which there appeared to be no post-mortem decomposition, were also necropsied. The investigators noted the main gross lesions observed in 103 necropsied pigs, of which 93 were euthanized.
Sick pigs were treated individually by injection of antibiotics, which included procaine penicillin (Depocillin; Intervet Canada Ltd, Whitby, Ontario, Canada) and florfenicol (Nuflor; Schering-Plough Animal Health, Pointe-Claire, Québec, Canada).
Statistical analysis
For the individual barn analysis, the two-sample proportions
test was used. For each of the two proportions being compared (one
vaccinate = Vp per barn and one control = Cp per barn), barn-level
mortality was used as the numerator and the total number of pigs
placed at the onset of the study was used as the denominator. The
null hypothesis tested was Vp = Cp. The starting P value
representing a significant difference between the two treatment
groups was P < .05. The Bonferroni adjustment was used to
calculate an adjusted significant P value level of
P < .0125 (.05 ÷ 4) for each test conducted on the data
from each of the four barns.
For the overall four-barn analysis, the one-way analysis of variance test was used. The four mortality rates were calculated for each treatment group as (number of deaths ÷ number of pigs placed), and the four values for the vaccinates and four values for the controls were compared. Homogeneity of treatment group variances was confirmed using Levene’s test. Differences between the vaccinate and control group mortality rates were considered statistically significant where P < .05. All statistical analyses were conducted in Statistix 9 (Analytical Software, Tallahassee, Florida).
Histological scoring
Ten tissues (cranial and caudal lung; tracheobronchial, mesenteric and superficial inguinal lymph nodes; tonsil; spleen; Peyer’s patches; liver; and kidney) from each of 101 pigs were evaluated for histological lesions suggestive of PCVAD, and for quantification of the PCV2 load by immunohistochemistry (IHC) staining. The score for histological lesions ranged from 0 (no lesions) to 3. Grade 1 lesions for all lymphoid organs were defined as a mild increase in histiocytic cells in all areas plus a mild reduction in lymphoid follicles and less obvious germinal centers. Grade 2 lesions were defined as obviously fewer lymphoid follicles in lymphoid and other visceral organs and more numerous areas of mixed mononuclear cell infiltrations in visceral organs. Grade 3 lesions were defined as total loss of lymphoid follicles in all tissues and large numbers of histiocytic cells effacing the paracortical T-cell zones, with very extensive histiocytic cell infiltrations in all organs, and with or without viral inclusion bodies in syncytial cells.
The score for antigenic load on IHC staining also ranged from 0 (no antigen) to 3. An antigen-load score of 1 was defined as mild positive staining of one to five cells per high-power field or an occasional germinal center with mild positive staining of central dendritic cells. At score 1, lung, liver, and kidney lesions were focal, small collections of lymphocytes, usually mixed with histiocytic cells. Only a few scattered cells were IHC-positive, usually < 1 cell per high-power field. An antigen-load score of 2 was defined as > 5 IHC-positive cells per high-power field or all germinal centers of lymphoid tissues IHC-positive. An antigen-load score of 3 was defined as extremely large numbers of IHC-positive cells in all tissues in high-power fields.
The pathologist who performed the evaluation (EC) was blinded to treatment.
Virological and serological investigations
Samples from a total of five pigs originating from three barns, showing gross lesions consistent with PCVAD, were submitted to a diagnostic laboratory (Faculté de Médecine Vétérinaire, Université de Montréal, St-Hyacinthe, Québec, Canada) for identification of PCV2 by polymerase chain reaction (PCR) and sequencing of the whole genome.
One hundred and twenty pigs (60 vaccinated, 60 controls) were ear-tagged, and blood samples were collected by puncture of the vena cava at days 1, 30, and 58 post vaccination. Twenty pigs vaccinated at each of 26, 40, and 52 days of age, ie, in three different nursery barns at the time of the first sampling, and 20 unvaccinated control pigs from the same age groups, were blood tested for PCV2 IgG antibodies. The tested pigs were randomly selected from a few pens of the nursery room housing each of these age groups. These pens were selected for convenience, ie, pens of vaccinates and controls that were close to each other.
Two serological tests were used: the Ingezim Circovirus IgG/IgM ELISA (Ingenasa, Madrid, Spain) and an indirect fluorescent antibody (IFA) test (Biovet Inc, St-Hyacinthe, Québec, Canada). For the ELISA test, the results were expressed in sample-to-positive ratios (S:P ratios), with samples considered positive at S:P ≥ 0.3. For the IFA, sera were either negative or positive at dilutions of 1 to 50, 1 to 200, 1 to 800, 1 to 3200, and 1 to 12,800.
Response to vaccination
Clinical and necropsy observations
No local or systemic reactions of any kind were noted either immediately after injection or in the days and weeks that followed. The mortality rate in the nursery pigs during the study was 0.4%, (six controls, nine vaccinates), which was similar to the rate prior to the study. The farm manager noticed no signs of disease while the pigs were in the nursery. Similarly, the investigator (RD) noted no abnormal signs during weekly visits while the pigs were in the nursery.
In this system, pigs historically began to show clinical signs of PCVAD approximately 3 to 4 weeks post placement in the finisher, with a range of 10 to 70 days post placement. During the trial, pigs began to show clinical signs of PCVAD approximately 3 weeks post placement (80 to 85 days of age) in all finishing units that took part in the study except in Barn 4, where signs of PCVAD were noticed approximately 1 week earlier. Clinical signs included wasting, lack of response to conventional treatments (ie, antimicrobial injections), pallor, and mortality. Table 1 shows mortality results (including pigs that died and those that were euthanized) for pigs vaccinated at different ages and control pigs placed in the four finishing units. For each of the finishing batches, mortality was significantly lower in vaccinated pigs than in controls.
Table 1: Mortality rates in controls and vaccinates in four finishing units on different sites in a commercial swine farm in eastern Canada*
* Nursery pigs were placed in the finisher units at
approximately 62 days of age. † Within farm (individual barn-level) two-sample proportions test. ‡ One-way analysis of variance. § Total number of pigs that died in the four barns divided by the total number of pigs that were introduced into these barns. |
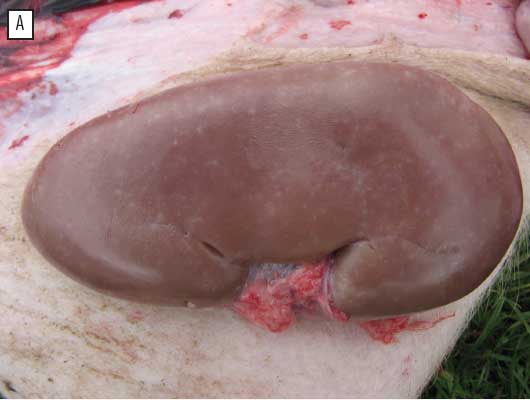
Overall, 86 control and 17 vaccinated pigs were necropsied. Numbers of control and vaccinated pigs differed because fewer vaccinated pigs became sick and debilitated, for which euthanasia was justified. Table 2 shows the frequency of the main gross lesions observed in necropsied pigs. The most frequent observation was enlarged lymph nodes. Various pneumonic lesions were observed (eg, cranio-ventral consolidation of lungs and lungs that did not collapse normally), but in an irregular pattern, and none of these lesions by themselves were sufficient to suggest PCVAD. The lungs of five control pigs showed marked interlobular edema, a lesion which has also been reported in cases of PCVAD.1 The kidneys of 25 pigs (23 controls and two vaccinates) had white spots or stains (Figure 1A). Among these 25 pigs, 22 had multiple tissues with scores of ≥ 2 for both histological lesions and PCV2 load (data not shown). One had multiple tissues with a histological score of ≥ 2, one had multiple tissues with a PCV2 load score of ≥ 2, and one had scores of 0 or 1 for both histological lesions and PCV2 load.
Table 2: Numbers of vaccinated and control pigs with various gross lesions at necropsy in the finisher*
* Multi-site system and vaccination protocol described in Table 1. Pigs from the four finishing units were selected for necropsy by the farm manager and the investigator on the basis of the likelihood that they would die, would be condemned at slaughter, or were suffering. |
||||||||||||||||||||||||||||
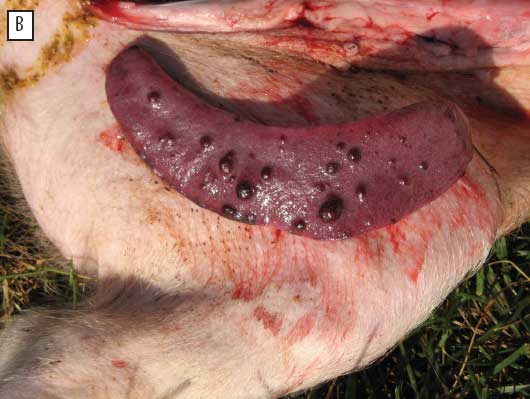
| Figure 1: Gross lesions of porcine circovirus associated
disease in a commercial swine-production system in eastern Canada:
(A) white spots on the kidney of a finisher pig, diagnosed
histologically as interstitial nephritis, and (B) splenic infarcts
in a finisher pig.
|
One vaccinate and two controls had very large and necrohemorrhagic spleens, four controls had splenic infarcts ranging from mild and in relatively small numbers to evident and numerous, and one control had a very large spleen as well as infarcts. Not all pigs with gross splenic lesions had high scores for histological lesions and PCV2 loads, but the pig with the most obvious and numerous infarcts (Figure 1B) had scores of 3 for all tissues examined by IHC and scores of 3 for histological lesions in eight of ten tissues. Two of the seven pigs in this study that had cutaneous lesions compatible with porcine dermatitis and nephropathy syndrome also had splenic infarcts.
While the stomach was not systematically examined in this study, 18 pigs (one vaccinate and 17 controls) had gastric ulcers. In most cases, the stomach was opened because of the marked anemia of the carcass. Thus, if every stomach had been opened to verify the presence or absence of an ulcer, the number of gastric ulcers observed might have been larger. The same is true for enteritis lesions: intestines were not systematically evaluated and were closely examined only when they appeared thickened on palpation, or when it appeared that the pig had suffered from diarrhea. None of the vaccinates, but 13 of the controls, had enteritis lesions ranging from simple thickening of the intestinal wall to necrosis. Some of these lesions could not be differentiated grossly from ileitis.
Histological lesions and viral loads
Table 3 shows the percentages of vaccinated and control pigs that had scores of ≥ 2 for histological lesions compatible with PCVAD, and for PCV2 antigenic load in the ten tissues examined. Eight of the 17 necropsied vaccinated pigs (47%) and two of the 84 necropsied control pigs (2%) had scores 0 or 1 for both evaluation methods and for the ten tissues examined. Thus, approximately half of the vaccinated pigs necropsied had died or been euthanized for causes other than PCVAD. In contrast, in almost all control pigs, the histological lesions and antigenic loads were compatible with a diagnosis of PCVAD. In Barns 1, 2, 3, and 4, the numbers of necropsied vaccinated pigs that had histological lesions and antigenic loads suggestive of PCVAD were four of six, zero of three, four of seven, and one of one, respectively.
Table 3: Number of necropsied vaccinates and controls with scores of ≥ 2 for histological lesions and PCV2 antigen loads in various tissues*
* Multi-site system and vaccination protocol described in Table 1. Selection of pigs for necropsy described in Table 2. Scores for histological lesions ranged from 0 (no lesions) to 3; scores for antigenic load on immunohistochemistry ranged from 0 (no antigen) to 3. † HE = histological staining of tissues with hematoxylin-eosin. ‡ IHC = detection of PCV2 antigen in tissues using immunohistochemistry. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The results obtained also suggested that there was little difference between inguinal, tracheobronchial, and mesenteric lymph nodes, in terms of detection of lesions and antigenic loads, and that lymph nodes, tonsils, and Peyer’s patches (ileum) produced more positive results than the other tissues tested.
Identification and sequencing of PCV2
Porcine circovirus type 2 was detected by PCR in all five pigs from which samples had been submitted for identification and sequencing. Sequences from one pig in Barn 3 and two pigs in Barn 4 were identified as PCV2b. Sequencing of the whole genome revealed that the three isolates were 99.1% to 99.4% homologous.
Serological results
Pigs that were serologically tested had been vaccinated at 26 days (finishing unit 3), 40 days (finishing unit 2), and 52 days of age (finishing unit 1). Approximately 60% and 80% of the pigs vaccinated at 26 days of age were seropositive on day 1 post vaccination by ELISA and IFA, respectively (data not shown).
The ELISA serological results indicated no detectable serocoversion following vaccination of pigs at 26 days of age. On day 30 post vaccination, only one of 20 and two of 20 controls and vaccinates, respectively, were seropositive, while nine of 16 and eight of 19, respectively, were seropositive on day 58 post vaccination. For pigs vaccinated at 40 or 52 days of age, there were numerically more vaccinated pigs seropositive on day 30 post vaccination (five of 20 vaccinates versus one of 19 controls and 16 of 19 vaccinates versus seven of 19 controls), while almost all pigs were seropositive on day 58 post vaccination (18 of 20 vaccinates versus 14 of 15 controls vaccinated at 40 days of age and 18 of 19 vaccinates versus 17 of 20 controls vaccinated at 52 days of age).
Using the IFA test, more vaccinated pigs were seropositive on day 30 post vaccination (10 of 20 vaccinates versus three of 20 controls, 17 of 20 vaccinates versus 11 of 19 controls, and 19 of 20 vaccinates versus 15 of 19 controls for pigs vaccinated at 26, 40, and 52 days of age, respectively). The geometric mean titers of the vaccinated pigs at day 30 post vaccination were not much higher than those of the controls, except for pigs vaccinated at 52 days of age, for which the titers were about five times higher than for controls (data not shown).
Discussion
As reported elsewhere,3-5 the vaccine used was both safe and effective. In all four barns, mortality rate was significantly lower in vaccinates than in controls. The most frequent gross lesions observed in necropsied pigs (enlarged lymph nodes, white spots on the kidneys or enlarged kidneys, and various pneumonia lesions) are among lesions reported to be suggestive of PCVAD.1 Some pigs had obvious splenic infarcts, which have long been considered almost pathognomonic of classical swine fever, a disease that does not occur in Canada. Few reports have associated splenic infarcts and PCVAD, but Carman et al6 did report this lesion in Ontario pigs with PCVAD. There are also reports of splenic infarcts associated with porcine dermatitis and nephropathy syndrome.1 An increase in the prevalence of pigs with gastric ulcers is frequently noted when PCVAD is a problem, which was true in this study.1 Jensen et al7 reported that intestinal lesions associated with PCVAD may be indistinguishable from ileitis lesions associated with Lawsonia intracellularis. Some pigs in the present study had lesions resembling ileitis. These pigs were not evaluated histologically specifically for ileitis, but they had lesion scores and viral loads compatible with PCVAD. Furthermore, ileitis was not a disease with which this system traditionally had problems, and no treatments for ileitis were instituted in any of the four study barns.
In this study, the highest percentage of lesions and antigen loads occurred in lymph nodes, tonsils, and ileum. No specific lymph node among the three that were tested (tracheobronchial, mesenteric, and superficial inguinal) appeared to be more affected than others.
A positive PCR result for PCV2 was obtained from all five pigs tested, and the sequences of the three that were tested revealed that these isolates were similar to those that had been identified in the majority of cases of PCVAD in eastern Canada.6
Using either the ELISA or the IFA test, there was no apparent correlation between detection of antibodies and protection in vaccinated pigs. Depending on the test used, pigs vaccinated at 26 days of age showed either no or weak seroconversion post vaccination, yet mortality in these pigs was much lower than in controls. These results are in agreement with those of Fenaux et al,8 who showed protection in pigs that had not seroconverted following administration of a test vaccine. Given the apparent lack of consistent or strong seroconversion following vaccination in the present study, the reasons that might explain the protection obtained remain hypothetical. There is a possibility that antibodies may have been produced that were not detectable by the tests used. Using the same vaccine, Fachinger et al3 showed that 4 and 8 weeks post vaccination, vaccinated pigs had significantly higher PCV2 geometric mean antibody titers than controls. They also showed that in vaccinated animals, the mean PCV2 viral load in serum (P < .0001) and the mean duration of viremia (P < .0001) were lower than in controls. While PCV2 levels in serum samples were not tested in the present study, PCV2 load in the tissues of vaccinated pigs was much lower than in controls. It is also possible that the pigs in this study were protected by cell-mediated immunity stimulated by vaccination. While certain types of antibodies might play a role in protection against PCV2, it may not be sufficient to confer total protection, as the virus can be found in the blood and tissues of pigs long after they have produced high levels of antibodies against it.9
It is also apparent from the results obtained that pigs could be effectively vaccinated in the presence of maternal immunity. At the time of vaccination, 60% and 80% of the pigs vaccinated at 26 days of age were seropositive by ELISA and IFA, respectively, due to maternal antibodies. These results are relatively similar to those obtained in 40 Canadian herds tested the same year.10 The percentage of seropositive pigs would thus be even higher in pigs vaccinated at 19 to 22 days of age, the group for which the efficacy appeared to be best, with a controls-to-vaccinates mortality ratio of 19 to 1 (7.6% versus 0.4%). In the study of Fachinger et al,3 the efficacy of vaccination was also compared for pigs that had low (< 1:1000) or high (> 1:1000) IFA titers at the time of vaccination. Gain in these two groups of pigs was not significantly different, indicating that in that study, the level of maternal antibody at the time of vaccination had no impact on growth performance of vaccinated pigs. Results obtained with two other vaccines have also shown that it was possible to effectively vaccinate pigs against PCV2 in the presence of passively acquired immunity.11,12
Finally, while the essential role of PCV2 in PCVAD appears to be less and less debated, it is still possible that unidentified factors or organisms might act as triggering factors for the disease to develop in the field. However, the results obtained in the present study, with a vaccine that is based solely on PCV2 antigen, suggest that when PCV2 is properly controlled by vaccination of nursery pigs, losses associated with PCVAD can be reduced or eliminated.
Implications
- A one-dose PCV2 vaccine is effective in reducing mortality associated with PCVAD.
- Under the conditions of this study, enlarged lymph nodes and white spots on the kidneys are among the most frequent gross lesions suggestive of PCVAD.
- Splenic infarcts, usually associated with classical swine fever, may also be found in cases of PCVAD.
- Under the conditions of this study, in pigs vaccinated with the tested PCV2 vaccine, seroconversion to PCV2 ELISA and IFA assays is not correlated with efficacy of the vaccine.
- PCV2 vaccine may be effectively used in the presence of maternal immunity.
Acknowledgement
The authors would like to thank André Broes (Biovet Inc, St-Hyacinthe, Québec, Canada) and Daniel Richard and Bertrand Dufresne (Boehringer Ingelheim Canada Ltd, Burlington, Ontario, Canada), for their greatly appreciated collaboration in this study.
References
1. Segalés J, Allan GM, Domingo M. Porcine circovirus diseases. An Health Res Rev. 2005;6:119–142.
2. Agriculture Canada. Recommended code of practice for care and handling of pigs. Communications Branch, Agriculture Canada, Ottawa, Ontario, Canada. 1984. Publication 1771/E.
3. Fachinger V, Bischoff R, Ben Jedidia S, Saalmüller A, Elbers K. The effect of vaccination against porcine circovirus type 2 in pigs suffering from porcine respiratory disease complex. Vaccine. 2008;26:1488–1499.
4. Kixmöller M, Ritzmann M, Eddicks M, Saalmüller A, Elbers K, Fachinger V. Reduction of PMWS-associated clinical signs and co-infections by vaccination against PCV2. Vaccine. 2008;26:3443–3451.
5. Cline G, Wilt V, Diaz E, Edler R. Efficacy of immunizing pigs against porcine circovirus type 2 at three or six weeks of age. Vet Rec. 2008;163:737–740.
6. Carman S, McEwen B, DeLay J, van Dreumel T, Lusis P, Cai H, Fairles J. Porcine circovirus-2 associated disease in swine in Ontario (2004 to 2005). Can Vet J. 2006;47:761–762.
7. Jensen TK, Vigre H, Svensmark B, Bille-Hansen V. Distinction between porcine circovirus type 2 enteritis and porcine proliferative enteropathy caused by Lawsonia intracellularis. J Comp Path. 2006;135:176–182.
8. Fenaux M, Opriessnig T, Halbur PG, Elvinger F, Meng XJ. A chimeric porcine circovirus (PCV) with the immunogenic capsid gene of the pathogenic PCV type 2 (PCV2) cloned into the genomic backbone of the nonpathogenic PCV1 induces protective immunity against PCV2 infection in pigs. J Virol. 2004;78:6297–6303.
*9. Opriessnig T, Madson DM, Prickett JR, Pogranichniy RM, Meng XJ, Halbur PG. Long-term effect of PCV2-infection and reinfection on PCV2-antibody and DNA profiles in pigs over time. 5th Int Symp Emerg Reemerg Pig Dis. Krakow, Poland. 2007;45.
*10. Cunningham GL, Desrosiers R, Sanford SE. Seroprevalence of PCV2 in Canadian swine herds. Proc IPVS. Durban, South Africa. 2008;1:12.
11. Opriessnig T, Patterson AR, Elsener J, Meng XJ, Halbur PG. Influence of porcine circovirus type 2 (PCV2) maternal antibodies on efficacy of PCV2 vaccination to protect pigs from experimental infection with PCV2. Clin Vacc Immunol. 2008;15:397–401.
*12. Roerink F, Sno M, Thijssen R, van Woensel P. Influence of maternally derived antibodies on the efficacy of vaccination against porcine circovirus. 5th Int Symp Emerg Reemerg Pig Dis. Krakow, Poland. 2007;117.
* Non-refereed references.